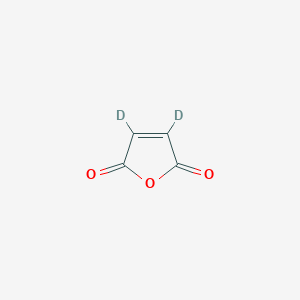
Maleic anhydride-d2
Overview
Description
Maleic anhydride-d₂ (C₄D₂O₃) is a deuterated derivative of maleic anhydride, where two hydrogen atoms are replaced with deuterium isotopes. This modification enhances its utility in analytical chemistry, particularly in nuclear magnetic resonance (NMR) spectroscopy, where it serves as an internal standard for quantifying compounds like 2,5-furandione . Structurally, maleic anhydride-d₂ retains the cyclic five-membered ring of maleic anhydride, with conjugated carbonyl groups enabling high reactivity in polymerization, copolymerization, and chemical derivatization . Its applications span polymer compatibilization (e.g., SEBS-g-MAH in polyamide blends ), wood modification for enhanced mechanical properties , and efficient succination of thiol compounds in synthetic chemistry .
Preparation Methods
Synthetic Routes and Reaction Conditions
Maleic anhydride-d2 can be synthesized through the deuteration of maleic anhydride. This process involves the exchange of hydrogen atoms with deuterium atoms. One common method is the catalytic exchange reaction using deuterium gas (D2) in the presence of a suitable catalyst. The reaction conditions typically involve elevated temperatures and pressures to facilitate the exchange process.
Industrial Production Methods
The industrial production of this compound follows similar principles to the production of maleic anhydride, with the additional step of deuteration. Maleic anhydride is typically produced by the vapor-phase oxidation of n-butane or benzene. The process involves passing the hydrocarbon through a catalyst bed at high temperatures in the presence of oxygen . The deuteration step can be integrated into this process by introducing deuterium gas during the oxidation reaction.
Chemical Reactions Analysis
Types of Reactions
Maleic anhydride-d2 undergoes various chemical reactions, including:
Hydrolysis: Reacts with water to form maleic acid-d2.
Esterification: Reacts with alcohols to form half-esters.
Diels-Alder Reactions: Acts as a dienophile in cycloaddition reactions.
Reduction: Can be reduced to form succinic anhydride-d2.
Substitution: Undergoes electrophilic addition and substitution reactions.
Common Reagents and Conditions
Hydrolysis: Water or hydroxide ions under mild conditions.
Esterification: Alcohols in the presence of acid catalysts.
Diels-Alder Reactions: Conjugated dienes under thermal conditions.
Reduction: Hydrogen gas in the presence of metal catalysts.
Substitution: Various electrophiles and nucleophiles under appropriate conditions.
Major Products Formed
Maleic acid-d2: from hydrolysis.
Half-esters: from esterification.
Cycloaddition products: from Diels-Alder reactions.
Succinic anhydride-d2: from reduction.
Scientific Research Applications
Polymer Chemistry
Unsaturated Polyester Resins
Approximately 50-55% of maleic anhydride production is directed towards unsaturated polyester resins (UPR). These resins are integral in creating fiberglass-reinforced plastics used in:
- Automobiles
- Boats
- Construction materials
- Electrical goods
Table 1: Applications of Maleic Anhydride in Polymer Chemistry
| Application | Description |
|---|---|
| Unsaturated Polyester Resins | Used in fiberglass composites for strength and durability |
| Alkyd Resins | Employed in coatings and paints for improved finish |
| Thermoplastic Polyurethanes | Derived from hydrogenated maleic anhydride for flexibility |
Pharmaceutical Applications
Maleic anhydride-d2 serves as a key intermediate in the synthesis of various pharmaceutical compounds. Its role in Diels-Alder reactions allows for the formation of complex molecular structures critical for drug development.
Case Study: Synthesis of Active Pharmaceutical Ingredients (APIs)
In a study published by the American Chemical Society, maleic anhydride was used to synthesize novel APIs through Diels-Alder reactions with dienes. This reaction facilitated the creation of compounds with enhanced bioactivity and specificity against targeted diseases .
Agricultural Chemicals
This compound is utilized in the formulation of agricultural chemicals, particularly as a precursor for insecticides and herbicides.
Table 2: Agricultural Applications
| Chemical Derived | Application |
|---|---|
| Malathion | Insecticide widely used in agriculture |
| Maleic Hydrazide | Growth regulator for crops |
Environmental Applications
The compound is also significant in environmental chemistry, particularly in water treatment processes. Maleic anhydride derivatives are employed to enhance the efficiency of surfactants used in wastewater treatment.
Case Study: Surfactants in Water Treatment
Research indicates that maleic anhydride-based surfactants improve oil removal efficiency from wastewater by enhancing emulsification properties . This application highlights the compound's role in promoting environmentally friendly practices.
Personal Care Products
This compound finds applications in personal care formulations, especially as a stabilizer and emulsifier in cosmetic products.
Table 3: Personal Care Applications
| Product Type | Role of this compound |
|---|---|
| Hair Sprays | Provides hold and texture |
| Skin Care Products | Acts as a delivery system for active ingredients |
Mechanism of Action
The mechanism of action of maleic anhydride-d2 involves its reactivity due to the electron-deficient conjugated double bond and the cyclic anhydride functionality. These features make it a versatile intermediate in various chemical reactions. In Diels-Alder reactions, for example, this compound acts as a dienophile, forming cycloaddition products through the interaction with conjugated dienes. The presence of deuterium atoms can influence the reaction kinetics and product distribution due to isotope effects .
Comparison with Similar Compounds
Succinic Anhydride
Structural Differences : Succinic anhydride (C₄H₄O₃) shares a linear four-carbon backbone with maleic anhydride but lacks the conjugated double bond, resulting in a saturated cyclic structure .
Reactivity and Applications :
- Succinic anhydride exhibits lower electrophilicity due to the absence of conjugation, making it less reactive in Diels-Alder reactions.
- It is preferred for mild acylation reactions, such as modifying amine-containing compounds, due to its slower hydrolysis rate compared to maleic anhydride .
- In succination efficiency tests, maleic anhydride outperforms succinic anhydride, achieving higher yields in thiol derivatization (e.g., 90% yield vs. 60% for succinic anhydride under identical conditions) .
Data Table 1: Comparative Succination Efficiency
| Compound | Reaction Efficiency (%) | Hydrolysis Stability |
|---|---|---|
| Maleic anhydride-d₂ | 90 | Low (rapid hydrolysis) |
| Succinic anhydride | 60 | Moderate |
| Maleic acid | 45 | High |
Phthalic Anhydride
Structural Differences : Phthalic anhydride (C₈H₄O₃) features an aromatic benzene ring fused to an anhydride group, contrasting with the aliphatic structure of maleic anhydride .
Reactivity and Applications :
- The aromatic ring enhances thermal stability, making phthalic anhydride suitable for high-temperature applications (e.g., plasticizer production).
- Maleic anhydride is more reactive in copolymerization (e.g., with vinyl acetate or styrene) due to its electron-deficient double bond, enabling robust hydrogel formation .
- Occupational exposure risks are similar for both compounds, with inhalation and dermal contact being primary pathways .
Maleic Acid
Structural Differences : Maleic acid (C₄H₄O₄) is the hydrolyzed form of maleic anhydride, containing two carboxylic acid groups instead of an anhydride ring .
Reactivity and Applications :
- Maleic anhydride hydrolyzes rapidly in aqueous environments, limiting its use in vivo, whereas maleic acid remains stable and is employed in cell culture studies .
- In polymer grafting, maleic anhydride forms covalent bonds with polyolefins (e.g., PP-g-MAH), while maleic acid primarily interacts via hydrogen bonding, resulting in weaker mechanical enhancements .
Fumaric Acid
Structural Differences: Fumaric acid (C₄H₄O₄) is a trans-isomer of maleic acid, with a non-conjugated double bond . Reactivity and Applications:
- Fumaric acid’s lower reactivity in succination reactions leads to by-product formation (e.g., thiol dimerization), whereas maleic anhydride achieves cleaner product profiles .
- Both compounds are intermediates in the tricarboxylic acid (TCA) cycle but diverge in industrial use; fumaric acid is favored in food additives, while maleic anhydride dominates polymer synthesis .
Maleimides
Structural Differences : Maleimides (e.g., C₄H₂O₂N-R) replace one oxygen atom in the maleic anhydride ring with an imide group .
Reactivity and Applications :
- Maleimides exhibit superior thermal stability and are widely used in epoxy resins and bioconjugation (e.g., antibody-drug conjugates).
- Maleic anhydride’s higher electrophilicity makes it more effective in nucleophilic additions (e.g., with thiourea derivatives) compared to maleimides .
Key Research Findings
- Antifungal Activity : Derivatives like 2,3-dimethylmaleic anhydride show potent antifungal activity against Sclerotinia sclerotiorum (EC₅₀ = 0.31 mM), outperforming succinic and phthalic anhydrides .
- Polymer Grafting : MAH grafting reduces polypropylene (PP) crystallinity by 30% but enhances compatibility in HDPE/PP blends, improving impact strength by 30-fold .
- Sustainable Production : Maleic anhydride can be synthesized from biomass-derived furans (e.g., HMF) using V-based catalysts, offering a renewable alternative to petroleum-based routes .
Biological Activity
Maleic anhydride-d2, a deuterated form of maleic anhydride, is a compound that has garnered attention for its biological activity, particularly in the fields of pharmacology and materials science. This article explores the biological activity of this compound, including its effects on cellular systems, potential therapeutic applications, and safety considerations.
Maleic anhydride (MA) is a cyclic anhydride of maleic acid, known for its reactivity and ability to form copolymers. The deuterated variant, this compound, is synthesized by replacing hydrogen atoms with deuterium. This modification can affect the compound's stability and reactivity, making it useful for specific applications in research and industry.
Antitumor Effects
Research has shown that maleic anhydride copolymers exhibit significant antitumor activity. For instance, studies on maleic anhydride-vinyl acetate copolymers demonstrated up to 68% tumor regression in K562 cellular cultures (human chronic myeloid leukemia) depending on the molecular weight of the copolymer used . This suggests that this compound may also possess similar antitumor properties due to its structural similarities.
Cytotoxicity
In vitro studies indicate that maleic anhydride copolymers can induce cytotoxicity in various cancer cell lines. The cytotoxic effects are often correlated with the molecular weight and composition of the copolymer. For example, copolymers with higher molecular weights showed increased cytotoxicity . This characteristic could be leveraged in developing targeted cancer therapies.
Respiratory Sensitization
Several case studies have documented adverse health effects associated with exposure to maleic anhydride. One notable case involved a worker who developed respiratory symptoms after exposure to maleic anhydride in a manufacturing setting. Symptoms included cough, breathlessness, and wheezing, which were linked to IgE-mediated allergic reactions . This highlights the importance of understanding the biological activity of this compound not only in therapeutic contexts but also concerning occupational health.
Genotoxicity Studies
Genotoxicity assessments have shown mixed results regarding maleic anhydride's potential to cause chromosomal aberrations. While some studies found no increase in chromosomal damage in animal models exposed to varying concentrations of maleic anhydride, others indicated possible genotoxic effects under certain conditions . This necessitates further investigation into the safety profile of this compound.
Summary of Key Studies
Q & A
Basic Research Questions
Q. What are the primary synthetic routes for preparing maleic anhydride-d₂, and how can isotopic purity be validated?
Maleic anhydride-d₂ (C₄D₂O₃) is synthesized via deuteration of maleic anhydride using deuterated reagents (e.g., D₂O or deuterated acids). Isotopic purity (>98% deuterium incorporation) is critical for applications in kinetic isotope effect studies or NMR spectroscopy. Validation methods include:
- Mass spectrometry (MS) to confirm molecular weight (100.07 g/mol) and isotopic distribution.
- ¹H NMR spectroscopy to detect residual proton signals, ensuring minimal non-deuterated impurities .
- FT-IR spectroscopy to compare vibrational modes (e.g., C=O stretching at ~1850 cm⁻¹) with non-deuterated analogs, noting shifts due to deuterium substitution .
Q. How should maleic anhydride-d₂ be handled to minimize hydrolysis during storage and experimentation?
Maleic anhydride-d₂ is hygroscopic and prone to hydrolysis, forming maleic acid-d₂. Methodological precautions include:
- Storing under inert gas (argon or nitrogen) in sealed, desiccated containers.
- Using anhydrous solvents (e.g., deuterated chloroform or THF-d₈) for reactions.
- Monitoring reaction environments for moisture via Karl Fischer titration .
Q. What are the key applications of maleic anhydride-d₂ in polymer chemistry research?
Maleic anhydride-d₂ is widely used as a deuterated monomer for:
- Copolymerization studies with styrene-d₈ to investigate reactivity ratios and sequence distribution via ²H NMR .
- Thermal degradation kinetics of deuterated polyimides, where isotopic labeling helps track degradation pathways using thermogravimetric analysis (TGA) coupled with mass spectrometry .
Advanced Research Questions
Q. How do solvent polarity and deuteration affect reactivity ratios in copolymerization with styrene?
Solvent effects on copolymerization (e.g., styrene/maleic anhydride-d₂) can lead to discrepancies in reactivity ratios (r₁, r₂). For example:
- In non-polar solvents (toluene), r₁ (styrene) ≈ 0.3 and r₂ (maleic anhydride-d₂) ≈ 0.05, favoring alternating copolymer formation.
- Polar solvents (DMF) may alter these ratios due to solvation effects on the transition state. Methodology : Use the Fineman-Ross (F-R) or Kelen-Tüdős (K-T) methods to calculate reactivity ratios from low-conversion data (<10%) to avoid composition drift .
Q. What are the challenges in analyzing kinetic isotope effects (KIEs) in Diels-Alder reactions using maleic anhydride-d₂?
Deuteration at the α-position (C₄D₂O₃) introduces measurable KIEs in Diels-Alder reactions:
- Experimental design : Compare reaction rates of maleic anhydride-d₂ vs. non-deuterated analogs under identical conditions (temperature, solvent).
- Data interpretation : Use Arrhenius plots to isolate isotopic effects on activation energy (Eₐ). For example, a study on bitumen modification showed Eₐ increased by 5–10% with deuteration, attributed to reduced vibrational entropy .
- Contradictions : Some studies report negligible KIEs due to competing mechanistic pathways, requiring DFT calculations to resolve .
Q. How can maleic anhydride-d₂ be used to trace degradation mechanisms in deuterated polyimides?
Deuterated polyimides synthesized from maleic anhydride-d₂ enable precise tracking of thermal degradation:
- Method : Perform pyrolysis-GC/MS on deuterated vs. non-deuterated polymers.
- Key findings : Deuterated polymers show delayed degradation onset (~10°C higher) due to stronger C-D bonds, confirmed by isotopic fragment analysis in MS .
Q. Data Contradiction Analysis
Q. Why do reported reactivity ratios (r₁, r₂) for styrene/maleic anhydride-d₂ copolymerization vary across studies?
Discrepancies arise from:
- Solvent effects : Polar solvents stabilize charge-separated transition states, altering r₁ and r₂ .
- Deuteration impact : Isotopic substitution subtly changes monomer electronic properties, affecting reactivity.
- Methodological bias : High-conversion data (>15%) skew ratios due to the "composition drift" phenomenon. Recommendation : Use low-conversion (<10%) data and the Yezrielev-Brokhina-Roskin (YBR) method for robust calculations .
Q. Methodological Best Practices
Q. What analytical techniques are optimal for characterizing deuterated copolymers derived from maleic anhydride-d₂?
- ²H NMR spectroscopy : Resolves sequence distribution and deuteration efficiency in copolymers (e.g., poly([styrene-d₈]-alt-[maleic anhydride-d₂])) .
- FT-IR spectroscopy : Monitors anhydride ring-opening during hydrolysis or post-polymerization modifications .
- Size-exclusion chromatography (SEC) : Coupled with multi-angle light scattering (MALS) to determine molecular weight distributions without isotopic interference .
Q. How can researchers mitigate side reactions during maleic anhydride-d₂ grafting onto polymers?
Common side reactions (e.g., homopolymerization or hydrolysis) are minimized by:
- Using microwave-assisted grafting to reduce reaction time and improve selectivity (e.g., grafting onto polypropylene films with >90% efficiency) .
- Adding free-radical inhibitors (e.g., hydroquinone) to suppress homopolymerization .
Properties
IUPAC Name |
3,4-dideuteriofuran-2,5-dione | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C4H2O3/c5-3-1-2-4(6)7-3/h1-2H/i1D,2D | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
FPYJFEHAWHCUMM-QDNHWIQGSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1=CC(=O)OC1=O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
[2H]C1=C(C(=O)OC1=O)[2H] | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C4H2O3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID80514711 | |
| Record name | (~2~H_2_)Furan-2,5-dione | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80514711 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
100.07 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
33225-51-3 | |
| Record name | (~2~H_2_)Furan-2,5-dione | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80514711 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Maleic anhydride-d2 | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details





Synthesis routes and methods III
Procedure details







Synthesis routes and methods IV
Procedure details





Synthesis routes and methods V
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.