Cobaltous nitrate hexahydrate
- Click on QUICK INQUIRY to receive a quote from our team of experts.
- With the quality product at a COMPETITIVE price, you can focus more on your research.
Overview
Description
Cobaltous nitrate hexahydrate (Co(NO₃)₂·6H₂O) is a red crystalline compound widely used in industrial and laboratory settings. Below are its key characteristics:
Mechanism of Action
Cobalt(II) nitrate hexahydrate, also known as Cobalt nitrate hexahydrate or Cobaltous nitrate hexahydrate, is a red-brown crystalline structure that is soluble in water and organic solvents . It is commonly used in the preparation of cobalt-based catalysts, pigments, chemicals, and vitamin B12 preparations .
Target of Action
The primary targets of Cobalt(II) nitrate hexahydrate are organic reactions where it acts as a catalyst . It is also used in the preparation of metallic cobalt and cobalt-based dyes and pigments .
Mode of Action
Cobalt(II) nitrate hexahydrate interacts with its targets by facilitating chemical reactions. For instance, it is used as a catalyst to synthesize 5-Carboxanilide-dihydropyrimidinone derivatives by the condensation of acetoacetanilides, aldehydes, and urea/thiourea . It also aids in the nitration of indoles using tert-butyl nitrite as the nitro source .
Biochemical Pathways
It is known to be involved in the synthesis of various cobalt-based compounds and catalysts .
Pharmacokinetics
It is known to be soluble in water and organic solvents, which suggests it could be readily absorbed and distributed in the body .
Result of Action
The molecular and cellular effects of Cobalt(II) nitrate hexahydrate’s action largely depend on its application. As a catalyst, it facilitates chemical reactions, leading to the formation of desired products . In the context of cobalt-based catalysts and pigments, it contributes to their synthesis .
Action Environment
The action, efficacy, and stability of Cobalt(II) nitrate hexahydrate can be influenced by various environmental factors. For instance, it is known to be hygroscopic, meaning it absorbs moisture from the environment . This property can affect its stability and efficacy. Furthermore, it is classified as an oxidizer, which means it can intensify fire . Therefore, it should be stored and handled carefully to prevent exposure to heat, sparks, open flames, and other ignition sources .
Biochemical Analysis
Biochemical Properties
Cobalt(II) nitrate hexahydrate is known to play a role in various biochemical reactions. It is commonly used in the preparation of cobalt-based catalysts, pigments, chemicals, and vitamin B12 preparations
Cellular Effects
It is known to be harmful if swallowed or inhaled, and it may cause an allergic skin reaction . It is also suspected of causing genetic defects .
Molecular Mechanism
It is known that the hexahydrate form consists of discrete [Co(OH₂)₆]²⁺ and [NO₃]⁻ ions . Each cobalt(II) atom is approximately octahedrally coordinated by six oxygen atoms, each from a different nitrate ion .
Temporal Effects in Laboratory Settings
In laboratory settings, Cobalt(II) nitrate hexahydrate is known to be stable under normal conditions . It is also known to be hygroscopic, meaning it absorbs moisture from the air
Dosage Effects in Animal Models
It is known to be harmful if swallowed or inhaled , and it may cause an allergic skin reaction . It is also suspected of causing genetic defects .
Metabolic Pathways
It is known to be used in the preparation of cobalt-based catalysts, pigments, chemicals, and vitamin B12 preparations .
Transport and Distribution
It is known to be soluble in water and other polar solvents , which suggests that it could be transported and distributed within cells and tissues via these solvents.
Subcellular Localization
Given its solubility in water and other polar solvents , it could potentially localize to any subcellular compartment that contains these solvents.
Scientific Research Applications
Catalysis
Cobaltous nitrate hexahydrate serves as a precursor for synthesizing cobalt-based catalysts. Its cobalt (II) ions can bind to organic ligands, forming complexes that exhibit specific catalytic properties. These complexes are particularly useful in:
- Organic Reactions : Cobaltous nitrate is utilized in reactions such as oxidation and hydrogenation due to its ability to stabilize reactive intermediates.
- Synthesis of Nanomaterials : It is employed in the production of cobalt nanoparticles, which have applications in catalysis and energy storage .
Material Synthesis
The compound is significant in the synthesis of materials with unique properties:
- Ceramics and Pigments : this compound is used in producing cobalt-based ceramics and pigments due to its vibrant color and thermal stability.
- Battery Components : It plays a role in developing cathode materials for lithium-ion batteries, enhancing their performance and stability .
Biological Studies
Cobalt is an essential trace element that plays a critical role in various biological processes. This compound is used in studies related to:
- Cellular Responses : Research indicates that cobalt ions can induce oxidative stress, leading to cellular damage or alterations in enzyme activities. For instance, exposure to cobaltous nitrate has been shown to cause cytotoxic effects in human neuroblastoma cells .
- Toxicology : Studies have highlighted its potential reproductive toxicity and carcinogenic properties, making it a subject of interest in toxicological research .
Environmental Science
This compound has applications in environmental studies:
- Soil Amendments : It can be used as a micronutrient fertilizer to enhance plant growth, particularly in cobalt-deficient soils.
- Pollution Studies : The compound's interactions with other environmental pollutants are studied to understand its behavior and impact on ecosystems .
Case Study 1: Catalytic Properties
Research conducted on this compound demonstrated its effectiveness as a catalyst for the oxidation of alcohols. The study found that by modifying the ligands attached to cobalt ions, researchers could tailor the catalytic activity for specific reactions, improving yields significantly.
Case Study 2: Biological Impact
A study examining the effects of cobaltous nitrate on human cell lines revealed that concentrations as low as 1 mM could induce significant cellular degeneration. This finding underscores the importance of understanding cobalt's biological effects for both health and environmental safety .
Comparison with Other Cobalt Compounds
| Compound Name | Chemical Formula | Key Characteristics |
|---|---|---|
| Cobalt(II) chloride | CoCl₂·6H₂O | Blue-green solid; used in dyeing and pigments. |
| Cobalt(II) sulfate | CoSO₄·7H₂O | Pink crystalline solid; used as a fertilizer. |
| Cobalt(II) acetate | Co(CH₃COO)₂·4H₂O | Used in organic synthesis; forms complexes easily. |
| Cobalt(II) carbonate | CoCO₃ | Occurs naturally; used for pigment production. |
This compound stands out due to its high solubility and oxidizing properties, making it particularly valuable for chemical synthesis compared to other less soluble or less reactive cobalt salts .
Q & A
Basic Questions
Q. What are the critical physical and chemical properties of cobaltous nitrate hexahydrate that influence experimental design?
- Key Properties :
- Solubility : 2170 g/L in water at 20°C (lower values, e.g., 1340 g/L, may reflect temperature or purity variations) .
- Melting Point : 55–57°C, with decomposition occurring above 74°C to cobalt oxide .
- pH : ~4.0 (20°C, 100 g/L aqueous solution) .
- Purity : Reagent-grade material often contains ≤0.005% trace metals (e.g., Ni, Fe, Cu), critical for minimizing interference in catalytic or spectroscopic studies .
Q. How should this compound be stored and handled to ensure safety and stability?
- Storage : Keep in airtight containers at 2–30°C to prevent deliquescence or hydrolysis .
- Safety Protocols :
- Use PPE (gloves, lab coats) to avoid skin contact, which may cause allergies or systemic toxicity (LD₅₀ = 691 mg/kg in rats) .
- In case of exposure, rinse skin/eyes with water for ≥15 minutes and seek medical attention .
- Decomposition : Avoid heating above 74°C in open air to prevent toxic NOₓ release .
Advanced Research Questions
Q. What synthetic strategies are effective for preparing cobalt-based catalysts using this compound?
- Impregnation Method : Dissolve in ethanol/water, load onto SBA-15 mesoporous silica, and calcine at 300–500°C to create Co₃O₄ nanoparticles for PET degradation catalysts .
- Hydrothermal Synthesis : Combine with nickel nitrate and graphene oxide in ethylene glycol to fabricate NiCo₂O₄ microspheres for supercapacitors, ensuring molar ratios are optimized for electrochemical performance .
- Quality Control : Use complexometric titration (99.0–102.0% assay) to confirm precursor purity, as trace Fe/Cu can alter catalytic activity .
Q. How can researchers resolve discrepancies in reported solubility and thermal stability data for this compound?
- Root Causes : Variations may arise from hydration state, measurement temperature, or impurities. For example, solubility at 20°C ranges from 1340 g/L to 2170 g/L due to differences in crystallinity or hygroscopicity.
- Experimental Mitigation :
- Standardize solution preparation by pre-equilibrating water temperature.
- Use thermogravimetric analysis (TGA) to monitor mass loss between 55°C (melting) and 100°C (decomposition) .
- Data Validation : Cross-reference with PubChem or HSDB entries, which consolidate multiple sources .
Q. What are the implications of this compound’s pH-dependent reactivity in coordination chemistry?
- Acidic Conditions : At pH < 4, the hexaaquacobalt(II) ion [Co(H₂O)₆]²⁺ dominates, favoring ligand substitution reactions for synthesizing complexes like [Co(NO₂)₆]³⁻ (used in K⁺ detection) .
- Neutral/Alkaline Conditions : Hydrolysis generates Co(OH)₂ precipitates; buffer solutions (e.g., ammonium acetate) stabilize intermediates during nanoparticle synthesis .
- Analytical Interference : Adjust pH to >6 when using EDTA titrations to avoid competing protonation effects .
Comparison with Similar Compounds
Chemical and Physical Properties
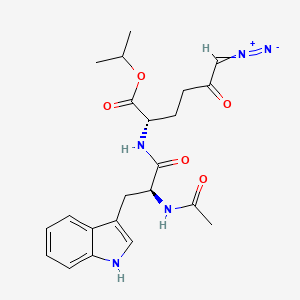
- Molecular Formula: Co(NO₃)₂·6H₂O
- Molecular Weight : 291.03 g/mol
- Melting Point : 55–57°C (decomposes above 74°C to cobalt oxide)
- Density : 1.87 g/cm³ at 20°C
- Solubility: 2170 g/L in water at 20°C; soluble in acetone and ethanol
- pH : 4.0 (100 g/L aqueous solution)
- Impurities : ≤0.001% Ni, ≤0.005% Cl⁻, ≤0.005% SO₄²⁻
Cobalt(II) Chloride Hexahydrate (CoCl₂·6H₂O)
Sodium Hexanitrocobaltat(III) (Na₃Co(NO₂)₆)
Magnesium Nitrate Hexahydrate (Mg(NO₃)₂·6H₂O)
*Estimated from general data.
Key Research Findings
- Thermal Stability : this compound decomposes to cobalt oxide at 74°C, limiting high-temperature applications compared to ZIFs (stable up to 550°C) .
- Regulatory Actions : Both cobaltous nitrate and cobalt chloride were withdrawn as feed additives in the EU due to toxicity concerns .
- Industrial Preference: Despite higher solubility, magnesium nitrate is favored in agriculture for dual nutrient (Mg²⁺, NO₃⁻) delivery, unlike cobaltous nitrate’s specialized industrial uses .
Properties
CAS No. |
10026-22-9 |
|---|---|
Molecular Formula |
CoH3NO4 |
Molecular Weight |
139.962 g/mol |
IUPAC Name |
cobalt;nitric acid;hydrate |
InChI |
InChI=1S/Co.HNO3.H2O/c;2-1(3)4;/h;(H,2,3,4);1H2 |
InChI Key |
YKPDYTLBLKQHDA-UHFFFAOYSA-N |
SMILES |
[N+](=O)([O-])[O-].[N+](=O)([O-])[O-].O.O.O.O.O.O.[Co+2] |
Canonical SMILES |
[N+](=O)(O)[O-].O.[Co] |
density |
1.88 g/cm³ |
melting_point |
55 °C |
Key on ui other cas no. |
23730-86-1 10026-22-9 |
physical_description |
RED CRYSTALS. |
Pictograms |
Corrosive; Irritant; Health Hazard |
solubility |
Solubility in water, g/100ml at 0 °C: 133.8 |
Synonyms |
cobalt nitrate cobaltous nitrate cobaltous nitrate dihydrate cobaltous nitrate dodecahydrate cobaltous nitrate hexahydrate cobaltous nitrate nonahydrate cobaltous nitrate tetrahydrate cobaltous nitrate trihydrate |
Origin of Product |
United States |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.