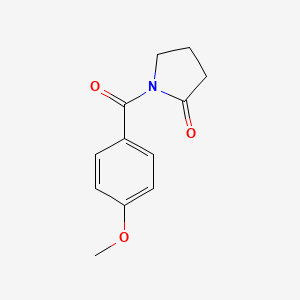
Aniracetam
Overview
Description
Aniracetam is a nootropic compound belonging to the racetam family, known for its cognitive-enhancing properties. It is chemically identified as 1-(4-methoxybenzoyl)-2-pyrrolidinone. This compound is primarily used to improve memory, learning, and attention, and has been studied for its potential benefits in treating cognitive impairments associated with cerebrovascular diseases and degenerative brain disorders .
Scientific Research Applications
Chemistry: Used as a model compound to study the effects of structural modifications on nootropic activity.
Biology: Investigated for its effects on neurotransmitter systems and synaptic plasticity.
Medicine: Studied for its potential to treat cognitive impairments, anxiety, and depression. .
Industry: Utilized in the development of new nootropic drugs and cognitive enhancers
Safety and Hazards
Aniracetam is considered toxic and contains a pharmaceutically active ingredient . Handling should only be performed by personnel trained and familiar with handling potent active pharmaceutical ingredients . It is advised to avoid breathing dust/fume/gas/mist/vapours/spray and avoid prolonged or repeated exposure .
Future Directions
While Aniracetam is effective at improving memory function in models of impaired memory, it has no effect in improving memory in healthy organisms . More recent, quality, and long-term research on this compound is necessary . It is conceivable that clinical trials will demonstrate beneficial effects of this compound in various disease states .
Mechanism of Action
Target of Action
Aniracetam primarily targets several receptors in the brain. These include the Glutamate receptor 2 , Glutamate receptor 3 , 5-hydroxytryptamine receptor 2A , and Dopamine D2 receptor . These receptors play crucial roles in cognitive functions, mood regulation, and memory processes .
Mode of Action
This compound acts on two main neurotransmitter systems in the brain: the glutamate and acetylcholine systems . By modulating the activity of receptors within these systems, this compound can enhance cognitive processes such as memory and learning . It also interacts with the cholinergic, dopaminergic, and serotonergic systems, which may mediate its wide range of anxiolytic properties .
Biochemical Pathways
This compound has been shown to positively modulate the AMPA receptor . It also increases cholinergic activity in the hippocampus, prefrontal cortex, and striatum . Furthermore, it protects against glutamate excitotoxicity . This compound may prevent the production and accumulation of Aβ by increasing α-secretase activity through two distinct pathways: increasing brain-derived neurotrophic factor expression and positively modulating metabotropic glutamate receptors .
Pharmacokinetics
This compound is rapidly and completely absorbed from the gastrointestinal tract . Its absolute systemic bioavailability is only about 02% . This compound has a high volume of distribution (2.5 L/kg), implying extensive extravascular distribution . It is very rapidly eliminated from the body, with a plasma elimination half-life of approximately 0.5 hours . This compound is completely metabolized, and the principal metabolites, N-anisoyl-γ-aminobutyric acid (N-anisoyl-GABA), 2-pyrrolidinone, succinimide, and anisic acid, are excreted via the urine (84%), the feces (2%), or as CO2 in expired air .
Result of Action
The molecular and cellular effects of this compound’s action are primarily related to its cognitive-enhancing properties. It improves memory and attention disturbances accompanying cerebrovascular diseases and degenerative brain disorders . This compound has been identified as a nootropic drug due to these memory effects . It also has modest effects similar to an anxiolytic .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of aniracetam typically involves the reaction of p-methoxybenzoic acid with pivaloyl chloride to form a mixed anhydride intermediate. This intermediate then reacts with 2-pyrrolidone to produce this compound. The reaction conditions include:
Step 1: p-Methoxybenzoic acid and pivaloyl chloride are mixed in dichloromethane with triethylamine under cooling conditions.
Step 2: The mixture is heated and refluxed, followed by the addition of 2-pyrrolidone.
Step 3: The reaction is stirred for 6-24 hours, then washed, dried, and crystallized to obtain this compound
Industrial Production Methods: The industrial production of this compound follows similar synthetic routes but is optimized for large-scale production. The process involves:
- Using readily available and cost-effective raw materials.
- Ensuring high reaction yield and product purity.
- Simplifying the operation to suit large-scale production .
Chemical Reactions Analysis
Types of Reactions: Aniracetam undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized to form its corresponding carboxylic acid derivative.
Reduction: Reduction reactions can convert this compound to its alcohol derivative.
Substitution: this compound can undergo nucleophilic substitution reactions, particularly at the carbonyl group.
Common Reagents and Conditions:
Oxidation: Common oxidizing agents include potassium permanganate and chromium trioxide.
Reduction: Reducing agents such as lithium aluminum hydride and sodium borohydride are used.
Substitution: Nucleophiles like amines and alcohols can react with this compound under acidic or basic conditions.
Major Products:
Oxidation: p-Anisic acid.
Reduction: this compound alcohol derivative.
Substitution: Various substituted this compound derivatives depending on the nucleophile used
Comparison with Similar Compounds
- Piracetam
- Oxiracetam
- Phenylpiracetam
- Pramiracetam
Aniracetam stands out due to its unique combination of cognitive and mood-enhancing effects, making it a versatile nootropic compound .
Properties
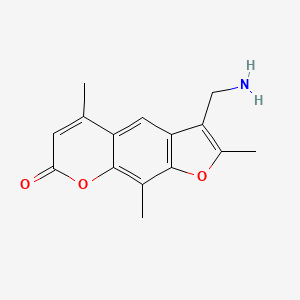
IUPAC Name |
1-(4-methoxybenzoyl)pyrrolidin-2-one | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C12H13NO3/c1-16-10-6-4-9(5-7-10)12(15)13-8-2-3-11(13)14/h4-7H,2-3,8H2,1H3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
ZXNRTKGTQJPIJK-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
COC1=CC=C(C=C1)C(=O)N2CCCC2=O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C12H13NO3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID5045128 | |
| Record name | Aniracetam | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5045128 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
219.24 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
72432-10-1 | |
| Record name | Aniracetam | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=72432-10-1 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Aniracetam [USAN:INN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0072432101 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Aniracetam | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB04599 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | aniracetam | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758223 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Aniracetam | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID5045128 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | ANIRACETAM | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/5L16LKN964 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Synthesis routes and methods I
Procedure details






Synthesis routes and methods II
Procedure details





Synthesis routes and methods III
Procedure details




Synthesis routes and methods IV
Procedure details








Synthesis routes and methods V
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
![3-[(2-Aminoethyl)dithio]propionic Acid](/img/structure/B1664882.png)