Osimertinib D6
Overview
Description
Osimertinib D6 is a compound under investigation for its ability to overcome resistance mechanisms in non-small cell lung cancer (NSCLC) driven by epidermal growth factor receptor (EGFR) mutations. Preclinical studies highlight its role in targeting EGFR variants with tertiary mutations, such as L858R/T790M/C797S, which confer resistance to third-generation EGFR tyrosine kinase inhibitors (TKIs) like osimertinib . In vitro experiments demonstrate that D6 promotes ubiquitination of the L858R/T790M/C797S-EGFR mutant protein, leading to reduced cell viability and proliferation in osimertinib-resistant (Os-R) PC-9 cell lines.
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of Osimertinib D6 involves the incorporation of deuterium atoms into the osimertinib molecule. This can be achieved through various methods, including:
Hydrogen-Deuterium Exchange: This method involves the replacement of hydrogen atoms with deuterium in the presence of a deuterium source, such as deuterium oxide or deuterated solvents.
Deuterated Reagents: Using deuterated reagents in the synthesis process can directly introduce deuterium atoms into the molecule.
Industrial Production Methods: Industrial production of this compound follows similar principles as the laboratory synthesis but on a larger scale. The process involves:
Large-Scale Hydrogen-Deuterium Exchange: Utilizing deuterium oxide or other deuterated solvents in large quantities.
Optimization of Reaction Conditions: Ensuring the reaction conditions are optimized for maximum yield and purity of the deuterated product.
Chemical Reactions Analysis
Types of Reactions: Osimertinib D6 undergoes various chemical reactions, including:
Oxidation: The compound can be oxidized to form various metabolites.
Reduction: Reduction reactions can modify the functional groups within the molecule.
Substitution: Substitution reactions can occur at specific sites within the molecule, leading to the formation of different derivatives.
Common Reagents and Conditions:
Oxidation Reagents: Common oxidation reagents include hydrogen peroxide and potassium permanganate.
Reduction Reagents: Sodium borohydride and lithium aluminum hydride are commonly used for reduction reactions.
Substitution Reagents: Halogenating agents and nucleophiles are used for substitution reactions.
Major Products Formed: The major products formed from these reactions include various metabolites and derivatives of this compound, which can have different pharmacological properties.
Scientific Research Applications
Efficacy in Clinical Studies
-
Preclinical Findings :
- Osimertinib D6 demonstrated significant anti-tumor activity against NSCLC cell lines expressing L858R/T790M-EGFR mutations. In studies, treatment with D6 at concentrations as low as 0.5 μM resulted in marked growth suppression and increased apoptosis markers such as cleaved PARP .
- Comparative studies indicated that D6 was less toxic to non-cancerous cells, suggesting a favorable therapeutic window .
-
Clinical Applications :
- In a pooled analysis of clinical data involving patients with uncommon EGFR mutations, osimertinib (and by extension, its derivatives) showed an overall response rate (ORR) of 48% and a disease control rate (DCR) of 86% . These findings underscore the potential for this compound to be effective even when conventional treatments fail.
- A systematic review highlighted that osimertinib significantly improved outcomes for patients with intracranial metastatic disease from NSCLC, showcasing its CNS penetration capabilities .
Case Studies
- Case Study 1 : A cohort study involving 90 patients treated with osimertinib revealed a 63.3% partial response rate and a median progression-free survival (mPFS) of 10.41 months. The incidence of adverse events was relatively low, with manageable side effects .
- Case Study 2 : Another analysis focused on patients with advanced lung adenocarcinoma demonstrated that osimertinib led to substantial improvements in overall survival compared to earlier therapies, reinforcing the efficacy of this treatment approach .
Comparative Data Table
Mechanism of Action
Osimertinib D6 exerts its effects by selectively inhibiting the epidermal growth factor receptor tyrosine kinase. It targets specific mutations in the epidermal growth factor receptor gene, including exon 19 deletions and exon 21 L858R mutations, as well as the T790M resistance mutation. By binding to these mutated receptors, this compound blocks the signaling pathways that promote cancer cell growth and survival.
Comparison with Similar Compounds
Comparative Analysis with Similar Compounds
Pharmacodynamic and Preclinical Comparisons
Table 1: Key Preclinical Parameters of EGFR-TKIs
| Compound | Target EGFR Mutations | IC50 (Resistant Cells) | BBB Penetrance | Resistance Overcome (C797S) |
|---|---|---|---|---|
| Osimertinib D6 | L858R/T790M/C797S | 5.15 µM (Os-R) | Not reported | Yes |
| Osimertinib | T790M, L858R, Ex19del | 3.466 µM (Os-R) | High | No |
| Almonertinib | T790M, L858R | Similar to osimertinib | Moderate | No |
| Afatinib | Ex19del, L858R | N/A | Low | No |
| Gefitinib/Erlotinib | Ex19del, L858R (1st-gen) | N/A | Low | No |
- Structural Similarity: Almonertinib shares a 94.87% structural similarity with osimertinib, differing only by a cyclopropyl group substitution on the pyrrole ring .
Clinical Efficacy and Pharmacokinetics
Table 2: Clinical Outcomes in EGFR-Mutant NSCLC
| Compound | Median PFS (Months) | Brain Metastasis Efficacy | OS Improvement vs. Chemotherapy |
|---|---|---|---|
| This compound | Under investigation | Not reported | Not reported |
| Osimertinib | 10.9–19.1 | Superior to afatinib | HR = 0.412 (vs. chemotherapy) |
| Afatinib | 11.0–13.7 | Limited | No significant difference |
| Platinum Chemotherapy | 4.0–5.3 | N/A | Reference |
- Brain Metastasis Control : Osimertinib achieves higher CNS exposure than afatinib, translating to longer median PFS in brain metastasis patients (19.1 vs. 13.7 months) . D6’s CNS efficacy remains unstudied.
- Post-Progression Outcomes : After osimertinib failure, chemotherapy provides a median PFS of 4 months, mirroring results from the MARIPOSA-2 trial .
Biological Activity
Osimertinib D6, a derivative of osimertinib, is a small-molecule compound designed to selectively inhibit the growth of tumor cells, particularly in non-small cell lung cancer (NSCLC) that exhibit resistance to earlier epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs). This article explores the biological activity of this compound, including its mechanisms of action, efficacy in overcoming drug resistance, and relevant case studies.
This compound targets the T790M mutation in the EGFR gene, which is a common mechanism of resistance to first- and second-generation EGFR TKIs. The compound selectively binds to the mutated form of EGFR, inhibiting its activity while sparing wild-type EGFR. This selectivity is crucial as it reduces systemic toxicity and enhances the therapeutic index.
- Inhibition of Tumor Growth : this compound has been shown to significantly suppress the growth of NSCLC cells harboring the T790M mutation. In vitro studies demonstrated that treatment with D6 at concentrations as low as 0.5 μM effectively inhibited colony formation in soft agar assays, indicating a strong anti-tumor effect .
- Induction of Apoptosis : Treatment with this compound resulted in a dose-dependent increase in cleaved PARP, a marker for apoptosis, in NCI-H1975 cells (EGFR T790M mutant). This suggests that D6 induces programmed cell death in resistant cancer cells .
- Protein Stability and Ubiquitination : The compound promotes the degradation of mutated EGFR through proteasomal pathways. Studies have shown that D6 increases levels of ubiquitinated EGFR proteins in NCI-H1975 cells, indicating enhanced turnover and degradation of the resistant mutant .
Efficacy Against Drug Resistance
This compound has demonstrated promising results in overcoming resistance to osimertinib itself. In preclinical models, D6 was effective at inhibiting tumor cell growth in lines resistant to osimertinib, suggesting its potential as a second-line therapy for patients who develop resistance after initial treatment .
Table 1: Efficacy Data for this compound
| Study Reference | Cell Line | Concentration (μM) | Effect on Growth | Induction of Apoptosis | Ubiquitination Increase |
|---|---|---|---|---|---|
| NCI-H1975 | 0.5 | Significant | Yes | Yes | |
| A549 (WT EGFR) | 0.5 | Minimal | No | No | |
| PC9 (Del19) | 0.5 | Minimal | No | No |
Clinical Case Studies
Several case studies highlight the clinical relevance of this compound:
- Case Study on Long-Term Survival : A patient with advanced lung adenocarcinoma who developed the T790M mutation after initial treatment with gefitinib was switched to osimertinib and subsequently to this compound after developing resistance. The patient achieved stable disease and maintained control over brain metastases for over four years .
- Efficacy in Combination Therapy : In combination with other agents like erlotinib, this compound has shown enhanced efficacy against resistant NSCLC models, indicating its potential role in combination therapy strategies .
- Clinical Trials : Ongoing clinical trials are evaluating the effectiveness of this compound in patients who have progressed on standard osimertinib therapy. Early results suggest improved outcomes compared to historical controls receiving no further treatment after progression .
Q & A
Basic Research Questions
Q. What are the key physicochemical and structural characterization methods for Osimertinib D6, and how do they differ from non-deuterated Osimertinib?
- Methodological Answer : Characterize this compound using nuclear magnetic resonance (NMR) spectroscopy to confirm deuterium incorporation at specific positions. Compare results with non-deuterated Osimertinib via high-resolution mass spectrometry (HRMS) and X-ray crystallography to assess isotopic effects on molecular structure. Include purity validation via HPLC with UV/Vis detection (≥98% purity threshold) . For reproducibility, document solvent systems, column specifications, and instrument calibration protocols in the supplementary materials .
Q. How to design a pilot study to evaluate this compound’s stability under varying pH and temperature conditions?
- Methodological Answer : Use forced degradation studies: incubate this compound in buffers (pH 1–10) at 25°C, 40°C, and 60°C for 24–72 hours. Monitor degradation via LC-MS/MS, quantifying parent compound loss and identifying degradation products. Include a negative control (non-deuterated Osimertinib) to isolate isotopic effects. Use kinetic modeling (e.g., Arrhenius equation) to extrapolate shelf-life .
Q. What in vitro models are appropriate for initial screening of this compound’s kinase inhibition profile?
- Methodological Answer : Employ recombinant EGFR (T790M/L858R mutant) kinase assays to measure IC50 values. Compare inhibition potency with non-deuterated Osimertinib using fluorescence resonance energy transfer (FRET) or radiometric assays. Include ATP concentration variations (1–10 mM) to assess competitive binding kinetics. Validate results with cell-based assays (e.g., Ba/F3 EGFR mutant cell lines) .
Advanced Research Questions
Q. How to resolve contradictions in reported metabolic stability data for this compound across preclinical studies?
- Methodological Answer : Conduct a systematic meta-analysis of existing data, stratifying studies by species (e.g., mouse vs. human hepatocytes), incubation conditions, and analytical methods. Use sensitivity analysis to identify variables (e.g., CYP enzyme activity, isotopic exchange rates) contributing to discrepancies. Validate findings via cross-laboratory reproducibility trials with standardized protocols .
Q. What experimental strategies can isolate the isotopic effect of deuterium in this compound on target binding and off-target toxicity?
- Methodological Answer : Perform molecular dynamics (MD) simulations to compare deuterated vs. non-deuterated Osimertinib’s binding kinetics to EGFR mutants. Validate computationally predicted differences using surface plasmon resonance (SPR) for binding affinity (KD) and transcriptomics (RNA-seq) to profile off-target gene expression in primary human hepatocytes .
Q. How to optimize a chiral separation protocol for this compound enantiomers during synthetic scale-up?
- Methodological Answer : Screen chiral stationary phases (CSPs) like cellulose tris(3,5-dimethylphenylcarbamate) using supercritical fluid chromatography (SFC). Optimize mobile phase composition (CO2/co-solvent ratio) and column temperature to achieve baseline separation (resolution >1.5). Validate enantiomeric purity via circular dichroism (CD) spectroscopy and replicate across three independent batches .
Q. Methodological Frameworks for Hypothesis Testing
Q. How to formulate a FINER (Feasible, Interesting, Novel, Ethical, Relevant) research question for this compound’s pharmacokinetic interactions?
- Methodological Answer :
- Feasible : Ensure access to deuterated compound (>95% isotopic purity) and human liver microsomes.
- Novel : Focus on unstudied drug-drug interactions (e.g., with CYP3A4 inducers).
- Ethical : Use in vitro models to minimize animal testing.
- Relevant : Align with FDA guidelines on deuterated drug development .
Q. What statistical approaches are robust for analyzing non-linear pharmacokinetic data from this compound studies?
- Methodological Answer : Apply non-compartmental analysis (NCA) for AUC and Cmax calculations, followed by nonlinear mixed-effects modeling (NONMEM) to account for inter-individual variability. Use bootstrapping (1,000 iterations) to estimate confidence intervals for deuterium-related metabolic shifts .
Q. Data Integrity and Reproducibility
Q. How to address batch-to-batch variability in this compound synthesis impacting experimental outcomes?
- Methodological Answer : Implement quality-by-design (QbD) principles: define critical synthesis parameters (e.g., deuterium source purity, reaction temperature), and use multivariate analysis (e.g., PCA) to correlate variability with biological activity. Share raw NMR and chromatographic data in public repositories (e.g., Zenodo) for transparency .
Q. What criteria should guide the selection of primary vs. secondary endpoints in this compound’s efficacy studies?
- Methodological Answer : Prioritize primary endpoints with direct clinical relevance (e.g., tumor volume reduction in PDX models) and secondary endpoints for mechanistic insight (e.g., phospho-EGFR levels). Pre-specify endpoints in a registered protocol (e.g., preclinicaltrials.eu ) to reduce bias .
Q. Integration with Existing Literature
Q. How to contextualize this compound’s preclinical data within the broader landscape of deuterated oncology agents?
- Methodological Answer : Conduct a systematic review using PRISMA guidelines to compare this compound’s pharmacokinetic (PK) parameters (e.g., t1/2, CL) with other deuterated drugs (e.g., deutetrabenazine). Highlight unique challenges, such as deuterium’s impact on metabolic enzyme kinetics, using comparative tables .
Q. What gaps in deuterated drug development warrant prioritization in future this compound research?
- Methodological Answer : Focus on understudied areas: (1) long-term isotopic effects on DNA repair mechanisms, (2) deuterium’s role in mitigating reactive metabolite formation, and (3) cost-benefit analysis of deuterated vs. non-deuterated drug manufacturing. Propose collaborative studies with isotopic labeling experts and regulatory bodies .
Properties
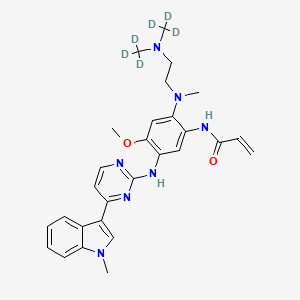
IUPAC Name |
N-[2-[2-[bis(trideuteriomethyl)amino]ethyl-methylamino]-4-methoxy-5-[[4-(1-methylindol-3-yl)pyrimidin-2-yl]amino]phenyl]prop-2-enamide | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C28H33N7O2/c1-7-27(36)30-22-16-23(26(37-6)17-25(22)34(4)15-14-33(2)3)32-28-29-13-12-21(31-28)20-18-35(5)24-11-9-8-10-19(20)24/h7-13,16-18H,1,14-15H2,2-6H3,(H,30,36)(H,29,31,32)/i2D3,3D3 | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
DUYJMQONPNNFPI-XERRXZQWSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CN1C=C(C2=CC=CC=C21)C3=NC(=NC=C3)NC4=C(C=C(C(=C4)NC(=O)C=C)N(C)CCN(C)C)OC | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
[2H]C([2H])([2H])N(CCN(C)C1=CC(=C(C=C1NC(=O)C=C)NC2=NC=CC(=N2)C3=CN(C4=CC=CC=C43)C)OC)C([2H])([2H])[2H] | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C28H33N7O2 | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
505.6 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Synthesis routes and methods I
Procedure details








Synthesis routes and methods II
Procedure details










Synthesis routes and methods III
Procedure details







Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.