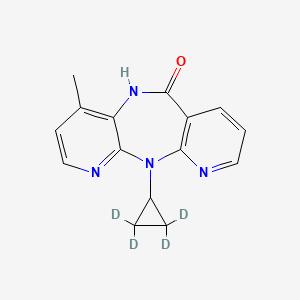
Nevirapine-D4
Overview
Description
Nevirapine-D4 is a deuterated analog of Nevirapine, a non-nucleoside reverse transcriptase inhibitor (NNRTI) used in HIV treatment. Deuterated compounds like this compound are designed to enhance pharmacokinetic properties, such as metabolic stability and half-life, by leveraging the kinetic isotope effect .
Nevirapine itself exists in anhydrous (C₁₅H₁₄N₄O, molecular weight 266.3) and hemihydrate (C₁₅H₁₄N₄O·½H₂O, molecular weight 275.3) forms . Its deuterated derivative would retain the core structure but with isotopic substitutions, likely in metabolically vulnerable positions (e.g., methyl or cyclopropyl groups).
Mechanism of Action
Target of Action
Nevirapine-D4, also known as 11-cyclopropyl-2,2,3,3-d4-5,11-dihydro-4-methyl-6H-dipyrido[3,2-b:2’,3’-e][1,4]diazepin-6-one, primarily targets the reverse transcriptase (RT) enzyme of the Human Immunodeficiency Virus Type 1 (HIV-1) . This enzyme plays a crucial role in the life cycle of the virus, as it transcribes viral RNA into DNA .
Mode of Action
This compound binds directly to the reverse transcriptase enzyme, causing a disruption of the enzyme’s catalytic site . This action blocks both the RNA-dependent and DNA-dependent DNA polymerase activities .
Biochemical Pathways
The primary biochemical pathway affected by this compound is the replication cycle of HIV-1 . By inhibiting the reverse transcriptase enzyme, this compound prevents the transcription of viral RNA into DNA . This action effectively halts the replication of the virus, thereby reducing viral load and slowing the progression of the disease .
Pharmacokinetics
It is readily absorbed (>90%) and freely partitions to all tissues, including the brain, due to its low level of protein binding . This compound is primarily metabolized in the liver through induction of CYP enzymes 3A4 and 2B6 . The metabolites are then excreted in the urine .
Result of Action
The primary molecular effect of this compound’s action is the inhibition of HIV-1 replication . On a cellular level, this results in a reduction of viral load and a slowing of disease progression . This compound is generally prescribed after the immune system has declined and infections have become evident .
Action Environment
The efficacy and stability of this compound can be influenced by various environmental factors. For instance, the presence of other antiretroviral drugs can impact the effectiveness of this compound . Additionally, patient-specific factors such as liver function can affect the metabolism and clearance of the drug .
Biochemical Analysis
Biochemical Properties
Nevirapine-D4 plays a crucial role in biochemical reactions, particularly in the inhibition of HIV-1 reverse transcriptase. This enzyme is essential for the replication of the HIV virus. This compound binds to the reverse transcriptase enzyme and inhibits its activity, preventing the transcription of viral RNA into DNA. This inhibition is non-competitive, meaning that this compound does not compete with the natural substrates of the enzyme. The interaction between this compound and reverse transcriptase involves binding to a specific site on the enzyme, causing a conformational change that reduces the enzyme’s activity .
Cellular Effects
This compound affects various types of cells, particularly those infected with HIV-1. By inhibiting the reverse transcriptase enzyme, this compound prevents the replication of the virus within infected cells. This leads to a reduction in viral load and helps in the management of HIV infection. Additionally, this compound can influence cell signaling pathways and gene expression related to viral replication and immune response. The inhibition of reverse transcriptase by this compound can lead to changes in cellular metabolism, as the cells are no longer producing viral components .
Molecular Mechanism
The molecular mechanism of this compound involves its binding to the reverse transcriptase enzyme. This binding disrupts the enzyme’s catalytic site, preventing it from carrying out its function of transcribing viral RNA into DNA. The disruption is caused by a conformational change in the enzyme, which reduces its activity. This compound does not compete with the natural substrates of the enzyme, making it a non-competitive inhibitor. This mechanism is crucial for its effectiveness in inhibiting HIV-1 replication .
Temporal Effects in Laboratory Settings
In laboratory settings, the effects of this compound can change over time. The stability of this compound is generally high, but it can degrade under certain conditions. Long-term studies have shown that this compound remains effective in inhibiting reverse transcriptase over extended periods. The degradation of this compound can lead to a reduction in its inhibitory activity. In vitro and in vivo studies have demonstrated that this compound can have long-term effects on cellular function, particularly in reducing viral replication and improving immune response .
Dosage Effects in Animal Models
The effects of this compound vary with different dosages in animal models. At lower doses, this compound effectively inhibits reverse transcriptase and reduces viral load without causing significant adverse effects. At higher doses, this compound can cause toxic effects, including hepatotoxicity and skin reactions. These adverse effects are dose-dependent and can be severe at very high doses. Animal studies have shown that there is a threshold dose below which this compound is safe and effective .
Metabolic Pathways
This compound is involved in several metabolic pathways, primarily through its interaction with cytochrome P450 enzymes. These enzymes metabolize this compound into various hydroxylated metabolites, which are then excreted from the body. The primary enzymes involved in the metabolism of this compound are CYP3A4 and CYP2B6. The metabolism of this compound can affect its efficacy and toxicity, as the metabolites may have different properties compared to the parent compound .
Transport and Distribution
This compound is transported and distributed within cells and tissues through passive diffusion and active transport mechanisms. It can cross cell membranes and accumulate in various tissues, including the liver, kidneys, and brain. The distribution of this compound is influenced by its lipophilicity and the presence of transport proteins. These proteins can facilitate the uptake and efflux of this compound, affecting its concentration in different tissues .
Subcellular Localization
The subcellular localization of this compound is primarily in the cytoplasm, where it interacts with the reverse transcriptase enzyme. This compound can also localize to other cellular compartments, such as the nucleus, where it may influence gene expression. The localization of this compound is influenced by its chemical properties and the presence of targeting signals that direct it to specific compartments. Post-translational modifications of this compound can also affect its localization and activity .
Biological Activity
Nevirapine-D4 is a deuterated analog of nevirapine, an antiretroviral drug used primarily in the treatment of HIV. This article explores its biological activity, including mechanisms of action, efficacy, safety profiles, and relevant case studies.
Nevirapine functions as a non-nucleoside reverse transcriptase inhibitor (NNRTI). By binding to the reverse transcriptase enzyme, it disrupts the viral replication process. The introduction of deuterium in this compound alters its pharmacokinetic properties, potentially enhancing its stability and reducing metabolic degradation compared to its non-deuterated counterpart.
Biological Activity and Efficacy
Research indicates that this compound exhibits similar efficacy to nevirapine in inhibiting HIV replication. However, studies have shown that the deuterated form may offer improved pharmacological properties:
- Stability : this compound has demonstrated increased metabolic stability, which may lead to prolonged therapeutic effects.
- Efficacy against Resistant Strains : Preliminary data suggest that this compound retains activity against certain HIV strains resistant to standard nevirapine therapy.
Case Studies and Clinical Findings
-
Plasma Concentration and Virologic Outcomes
A study involving 233 HIV patients receiving nevirapine-based ART revealed a correlation between plasma concentrations of nevirapine and clinical outcomes. Patients with suboptimal levels (<3400 ng/mL) exhibited higher rates of virologic failure and were more likely to harbor drug-resistant strains .Plasma Concentration (ng/mL) Virologic Failure Rate (%) Drug Resistance Mutations (%) <3400 60.5 16.3 3400 - 6000 30.2 9.9 >6000 10.0 3.5 -
Impact on CD8+ T Cell Function
Research highlighted that nevirapine can inhibit the anti-HIV activity of CD8+ T cells, which are crucial for immune response against HIV. In co-culture experiments, exposure to nevirapine at concentrations as low as 10 µM significantly reduced the antiviral function of CD8+ cells from long-term survivors . This finding underscores the importance of monitoring drug levels to mitigate potential immunosuppressive effects.
Safety Profile
This compound's safety profile remains under investigation. However, studies on nevirapine have reported side effects including:
- Rash : A common adverse effect that can range from mild to severe.
- Hepatotoxicity : Liver function should be monitored due to potential toxicity.
- Drug Interactions : Caution is advised when co-administering with other medications due to metabolic interactions.
Research Findings Summary
- This compound shows promise as an effective alternative in HIV treatment regimens.
- Its pharmacokinetic advantages may improve patient adherence and treatment outcomes.
- Ongoing studies are necessary to fully elucidate its safety and efficacy compared to traditional nevirapine.
Scientific Research Applications
Pharmacokinetics and Drug Monitoring
Nevirapine is commonly used in ART regimens for individuals living with HIV. The introduction of Nevirapine-D4 allows for more precise pharmacokinetic studies due to its distinct isotopic labeling. This enables researchers to differentiate between the administered drug and its metabolites in biological samples.
Methodologies for Quantification
Liquid chromatography-tandem mass spectrometry (LC-MS/MS) is the primary method employed for quantifying nevirapine and its metabolites. Recent studies have developed sensitive methods for detecting nevirapine levels in various biological matrices, including hair, plasma, and meconium.
- Hair Analysis : A study demonstrated the simultaneous determination of nevirapine and its metabolites (2-hydroxynevirapine and 3-hydroxynevirapine) in hair samples. This method involved liquid-liquid extraction followed by LC-MS/MS analysis. The results indicated that higher concentrations of nevirapine correlated with better adherence to ART among patients .
- Plasma and Meconium Studies : Other studies have focused on quantifying nevirapine in plasma and meconium. For instance, a method was validated for 16 antiretroviral drugs in meconium, showing high extraction efficiencies and linearity across a range of concentrations .
Case Studies Highlighting Applications
- Adherence Assessment : A notable case study assessed long-term adherence to ART by analyzing hair samples from HIV patients. The study found that patients with high adherence had significantly higher concentrations of nevirapine and its metabolites compared to those with low adherence. This finding underscores the potential of hair analysis as a reliable measure of drug adherence over time .
- Pharmacokinetic Profiles : In pharmacokinetic studies involving baboons, researchers utilized this compound to track the metabolism of nevirapine. The study provided insights into how the drug is processed in vivo, contributing valuable data for understanding its efficacy and safety profiles in humans .
Comparative Data Tables
The following table summarizes key findings from recent studies on nevirapine quantification methods:
| Compound | Matrix | Method | Limit of Quantification (pg/mg) | Recovery (%) | Linear Range (pg/mg) |
|---|---|---|---|---|---|
| Nevirapine | Hair | LC-MS/MS | 6 | 91.1 - 113.7 | 6 - 5000 |
| Nevirapine | Plasma | LC-MS/MS | 10 | 93.1 - 108.3 | 10 - 40000 |
| Nevirapine | Meconium | LC-MS/MS | 10 | 80.3 - 108.3 | 10 - 25000 |
Chemical Reactions Analysis
Metabolic Activation and Reactive Intermediate Formation
Nevirapine undergoes hepatic metabolism to form reactive intermediates, a pathway critical to understanding its toxicity. While Nevirapine-D4 retains the core structure, isotopic substitution (deuterium at four positions) may influence reaction kinetics due to the kinetic isotope effect.
Key Metabolic Pathways:
-
Oxidation : CYP3A4 and CYP2B6 mediate hydroxylation at positions 2, 3, 8, and 12, producing hydroxylated metabolites (e.g., 12-hydroxy-NVP) .
-
Sulfonation : 12-hydroxy-NVP is sulfonated to 12-sulfoxy-NVP, a reactive electrophile that forms DNA adducts (e.g., N3-NVP-dT and O4-NVP-dT ) .
-
Glucuronidation : Phase II metabolism via UGT enzymes (e.g., UGT2B7) facilitates excretion .
Isotope Effects in Metabolism:
Deuterium substitution at metabolically active positions (e.g., C12) could slow hydroxylation rates, potentially reducing reactive metabolite generation. This hypothesis aligns with studies on deuterated analogs designed to mitigate toxicity .
DNA and Protein Adduct Formation
This compound’s reactive metabolites covalently bind to nucleophilic sites in DNA and proteins, a process mirrored in non-deuterated nevirapine:
-
Biomimetic Studies : Reaction of 12-mesyloxy-NVP (a synthetic surrogate for 12-sulfoxy-NVP) with DNA produces N3-NVP-dT as the primary adduct, identified via LC-MS and synthetic standards .
-
Cytotoxicity : These adducts are implicated in mutagenesis and hepatotoxicity .
Analytical Data for this compound:
| Parameter | This compound | Nevirapine |
|---|---|---|
| Retention Time | 7.84 min | 7.84 min |
| Calibration Range | 11–50,000 pg/mg | 11–50,000 pg/mg |
| LOD | 3 pg/mg | 3 pg/mg |
| LOQ | 11 pg/mg | 11 pg/mg |
Data adapted from LC-MS/MS methods used to quantify nevirapine and metabolites in hair samples .
Reactivity with Oxidizing Agents
Nevirapine reacts with sodium hypochlorite (NaOCl), a common disinfectant, forming chlorinated byproducts. Deuterated analogs like this compound exhibit similar reactivity but may show altered kinetics:
-
Reaction Reversibility : Addition of reducing agents (e.g., sodium thiosulfate) reverses chlorination, suggesting non-covalent interactions dominate under certain conditions .
-
pH Dependence : Reaction rates increase at basic pH (e.g., pH 8), with complete Nevirapine depletion within 1 minute at 500 µM NaOCl .
Stability and Degradation Pathways
This compound demonstrates comparable stability to nevirapine under physiological conditions. Key degradation pathways include:
-
Hydrolysis : Labile amide bonds in the cyclopropylamino group may undergo cleavage under acidic conditions .
-
Photodegradation : Limited data exist, but structural analogs suggest susceptibility to UV-induced degradation.
Pharmacokinetic Interactions
While this compound itself is not therapeutic, its pharmacokinetics inform drug monitoring:
Q & A
Basic Research Questions
Q. What are the key physicochemical properties of Nevirapine-D4, and how do they influence experimental design in pharmacokinetic studies?
this compound, a deuterated analog of nevirapine, exhibits altered metabolic stability due to deuterium substitution at specific positions. Key properties include molecular weight (267.3 g/mol), logP (~1.8), and deuterium isotope effects (kH/kD ~2–5). Researchers should prioritize LC-MS/MS for quantification due to its isotopic differentiation capabilities. Experimental designs must account for solvent deuterium exchange and validate analytical methods using deuterated internal standards to minimize matrix effects .
Q. What validated methodologies exist for synthesizing this compound with high isotopic purity?
Synthesis typically involves deuterium exchange reactions under acidic or basic conditions, using deuterated reagents (e.g., D2O, CD3OD). Isotopic purity (>98%) is confirmed via <sup>1</sup>H-NMR (absence of non-deuterated proton signals) and high-resolution mass spectrometry. Researchers should optimize reaction kinetics to minimize byproducts and validate purity using orthogonal techniques (e.g., IR spectroscopy) .
Q. How does this compound’s deuterium substitution impact its binding affinity to cytochrome P450 enzymes compared to non-deuterated nevirapine?
Deuterium substitution reduces metabolic clearance by stabilizing C-H bonds at deuterated positions, increasing half-life. Competitive inhibition assays (e.g., CYP3A4/CYP2B6) using fluorogenic substrates and recombinant enzymes are recommended. Data should be analyzed via Michaelis-Menten kinetics, comparing Ki values between this compound and nevirapine to quantify isotope effects .
Advanced Research Questions
Q. How can researchers resolve contradictions in reported this compound efficacy data across in vitro vs. in vivo models of HIV resistance?
Discrepancies often arise from differences in metabolic enzyme expression (e.g., CYP3A4 in hepatocytes vs. systemic clearance in vivo). Advanced approaches include:
- Cross-model validation : Compare IC50 values in primary human hepatocytes with pharmacokinetic data from rodent models.
- Mechanistic modeling : Use physiologically based pharmacokinetic (PBPK) models to simulate deuterium effects on tissue distribution.
- Data reconciliation : Apply Bayesian statistics to integrate conflicting datasets, adjusting for covariates like protein binding and efflux transporter activity .
Q. What experimental strategies mitigate deuterium-related artifacts in this compound studies, particularly in long-term toxicity assays?
- Isotopic stability testing : Monitor deuterium loss via mass spectrometry under experimental conditions (e.g., elevated temperature, pH extremes).
- Control groups : Include non-deuterated nevirapine and vehicle controls to distinguish isotope-specific effects from baseline toxicity.
- Metabolite profiling : Use HR-MS/MS to identify deuterium-retained vs. lost metabolites, correlating findings with histopathological outcomes .
Q. How should researchers design studies to assess this compound’s potential for inducing viral resistance in combination therapies?
- Stepwise resistance selection : Expose HIV-1 strains (e.g., NL4-3) to subtherapeutic this compound concentrations over multiple passages.
- Genotypic/phenotypic analysis : Sequence reverse transcriptase mutations (e.g., K103N, Y181C) and quantify fold-changes in EC50 using plaque reduction assays.
- Synergy testing : Evaluate combination indices with tenofovir/efavirenz using the Chou-Talalay method to identify additive or antagonistic effects .
Q. What ethical and methodological considerations apply to clinical trials involving this compound in pediatric populations?
- Informed consent : Adapt protocols for guardian assent and age-appropriate participant communication.
- Dosing adjustments : Use allometric scaling based on body surface area and population PK modeling to account for developmental metabolic differences.
- Safety monitoring : Implement DSMBs (Data Safety Monitoring Boards) to review adverse events, particularly hepatic enzyme elevations. Reference NIH pediatric guidelines for preclinical-to-clinical translation .
Q. Methodological Frameworks
Q. Which statistical approaches are optimal for analyzing non-linear pharmacokinetic data from this compound studies?
- Non-compartmental analysis (NCA) : Calculate AUC, Cmax, and t1/2 using WinNonlin or Phoenix.
- Non-linear mixed-effects modeling (NLME) : Employ Monolix or NONMEM to estimate inter-individual variability and covariate effects.
- Bootstrap validation : Assess parameter uncertainty with 1,000 iterations to ensure robustness .
Q. How can FAIR data principles be implemented in this compound research to enhance reproducibility?
- Metadata standardization : Use ISA-Tab formats for experimental workflows and link datasets to unique identifiers (e.g., DOI).
- Repository integration : Deposit raw spectra, chromatograms, and PK curves in domain-specific repositories (e.g., ChEMBL, Zenodo).
- Peer review checks : Adopt NFDI4Chem standards for data quality assessment during manuscript submission .
Q. Contradiction and Innovation
Q. What novel applications of this compound are emerging in non-HIV research contexts, such as oncology or neurodegenerative diseases?
Recent studies explore its off-target kinase inhibition (e.g., EGFR T790M) and neuroprotective effects via NMDA receptor modulation. Researchers should:
Comparison with Similar Compounds
Structural and Functional Analogues
Nevirapine vs. Nevirapine-Related Compound A
- Nevirapine : C₁₅H₁₄N₄O; CAS 129618-40-2 (anhydrous) or 220988-26-1 (hemihydrate). Acts as an NNRTI by binding to HIV-1 reverse transcriptase .
- Nevirapine-Related Compound A : C₁₄H₁₄N₄O; CAS unspecified. A structural analog lacking the cyclopropyl group, which may reduce binding affinity or antiviral activity .
| Property | Nevirapine | Nevirapine-Related Compound A |
|---|---|---|
| Molecular Formula | C₁₅H₁₄N₄O | C₁₄H₁₄N₄O |
| Molecular Weight | 266.3 (anhydrous) | 254.29 |
| Solubility | Insoluble in water | Not reported |
| Pharmacological Role | NNRTI | Likely inactive metabolite |
Deuterated Compounds: N-Nitrosodibenzylamine-d4 and (R,S)-N-Nitrosoanabasine-d4
- N-Nitrosodibenzylamine-d4: C₁₄H₁₄N₂O; CAS 20002-23-6. A deuterated nitrosamine with unclassified health hazards but unknown toxicological properties .
- (R,S)-N-Nitrosoanabasine-d4 : C₁₀H₁₀D₄N₂O; CAS 1020719-68-8. A deuterated tobacco-specific nitrosamine, used as a reference standard in analytical chemistry .
| Property | Nevirapine-D4 (Inferred) | N-Nitrosodibenzylamine-d4 | (R,S)-N-Nitrosoanabasine-d4 |
|---|---|---|---|
| Deuterium Substitution | 4 H → ²H | 4 H → ²H | 4 H → ²H |
| Molecular Weight | ~270.3 (estimated) | 242.3 | 174.2 (C₁₀H₁₀D₄N₂O) |
| Stability | Enhanced metabolic | Unknown | Light-sensitive |
| Regulatory Status | Not classified | Unclassified hazards | Requires tight container |
Properties
IUPAC Name |
7-methyl-2-(2,2,3,3-tetradeuteriocyclopropyl)-2,4,9,15-tetrazatricyclo[9.4.0.03,8]pentadeca-1(11),3,5,7,12,14-hexaen-10-one | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C15H14N4O/c1-9-6-8-17-14-12(9)18-15(20)11-3-2-7-16-13(11)19(14)10-4-5-10/h2-3,6-8,10H,4-5H2,1H3,(H,18,20)/i4D2,5D2 | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
NQDJXKOVJZTUJA-CQOLUAMGSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=C2C(=NC=C1)N(C3=C(C=CC=N3)C(=O)N2)C4CC4 | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
[2H]C1(C(C1([2H])[2H])N2C3=C(C=CC=N3)C(=O)NC4=C(C=CN=C42)C)[2H] | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C15H14N4O | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
270.32 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details






Synthesis routes and methods III
Procedure details






Synthesis routes and methods IV
Procedure details





Synthesis routes and methods V
Procedure details




Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.