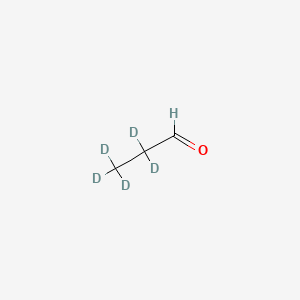
Propionaldehyde-2,2,3,3,3-d5
Overview
Description
Propionaldehyde-2,2,3,3,3-d5, also known as 2,2,3,3,3-pentadeuteriopropanal, is a deuterium-labeled compound. It is a stable isotope-labeled analog of propionaldehyde, where five hydrogen atoms are replaced by deuterium atoms. This compound is primarily used in scientific research as a tracer in metabolic studies and in the analysis of chemical reactions.
Scientific Research Applications
Propionaldehyde-2,2,3,3,3-d5 is widely used in scientific research due to its stable isotope labeling. Some of its applications include:
Metabolic Studies: Used as a tracer to study metabolic pathways and enzyme kinetics.
NMR Spectroscopy: Utilized in nuclear magnetic resonance (NMR) spectroscopy to study molecular structures and dynamics.
Pharmaceutical Research: Employed in the development of deuterium-labeled drugs to improve their metabolic stability and pharmacokinetics.
Environmental Analysis: Used in the analysis of environmental samples to trace the sources and pathways of pollutants.
Safety and Hazards
Mechanism of Action
Target of Action
Propionaldehyde-2,2,3,3,3-d5 is a stable isotope-labelled compound
Mode of Action
As a stable isotope-labelled compound, it behaves similarly to its non-labelled counterpart, but its pathway can be traced more accurately due to the presence of the deuterium atoms .
Biochemical Pathways
As a stable isotope-labelled compound, it is often used in metabolic studies to understand the biochemical pathways of the non-labelled compound .
Pharmacokinetics
As a stable isotope-labelled compound, it is often used in pharmacokinetic studies to understand the adme properties of the non-labelled compound .
Result of Action
As a stable isotope-labelled compound, it is often used in scientific research to study the effects of the non-labelled compound at the molecular and cellular level .
Action Environment
As a stable isotope-labelled compound, it is often used in environmental studies to understand the behavior of the non-labelled compound in various environmental conditions .
Biochemical Analysis
Biochemical Properties
It’s hypothesized that it could be used to investigate the metabolic pathways of propionate, a three-carbon fatty acid found in food and produced by gut bacteria. The specific enzymes and proteins it interacts with are yet to be identified.
Molecular Mechanism
Metabolic Pathways
Propionaldehyde-2,2,3,3,3-d5 could potentially be involved in the metabolic pathways of propionate. The specific enzymes or cofactors it interacts with, and its effects on metabolic flux or metabolite levels, are yet to be identified.
Preparation Methods
Synthetic Routes and Reaction Conditions
Propionaldehyde-2,2,3,3,3-d5 can be synthesized through the deuteration of propionaldehyde. One common method involves the exchange of hydrogen atoms with deuterium using deuterium oxide (D2O) in the presence of a catalyst. The reaction typically occurs under mild conditions to ensure the selective incorporation of deuterium atoms.
Industrial Production Methods
Industrial production of this compound involves the hydroformylation of ethylene with deuterium-labeled carbon monoxide and deuterium gas. This process yields deuterium-labeled propionaldehyde, which is then purified to obtain the desired compound .
Chemical Reactions Analysis
Types of Reactions
Propionaldehyde-2,2,3,3,3-d5 undergoes various chemical reactions typical of aldehydes, including:
Oxidation: It can be oxidized to propionic acid-2,2,3,3,3-d5 using oxidizing agents such as potassium permanganate or chromium trioxide.
Reduction: It can be reduced to propanol-2,2,3,3,3-d5 using reducing agents like sodium borohydride or lithium aluminum hydride.
Condensation: It can participate in aldol condensation reactions to form larger molecules.
Common Reagents and Conditions
Oxidation: Potassium permanganate (KMnO4) in an acidic medium.
Reduction: Sodium borohydride (NaBH4) in methanol or lithium aluminum hydride (LiAlH4) in ether.
Condensation: Base catalysts such as sodium hydroxide (NaOH) or acid catalysts like sulfuric acid (H2SO4).
Major Products
Oxidation: Propionic acid-2,2,3,3,3-d5.
Reduction: Propanol-2,2,3,3,3-d5.
Condensation: β-hydroxy aldehydes or ketones, depending on the reactants used.
Comparison with Similar Compounds
Similar Compounds
Propionaldehyde: The non-deuterated analog of Propionaldehyde-2,2,3,3,3-d5.
Acetaldehyde-d4: A deuterium-labeled analog of acetaldehyde.
Butyraldehyde-d7: A deuterium-labeled analog of butyraldehyde.
Uniqueness
This compound is unique due to its specific deuterium labeling, which provides distinct advantages in research applications. The presence of five deuterium atoms allows for precise tracking and analysis in metabolic studies and NMR spectroscopy, making it a valuable tool in scientific research .
Properties
IUPAC Name |
2,2,3,3,3-pentadeuteriopropanal | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C3H6O/c1-2-3-4/h3H,2H2,1H3/i1D3,2D2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
NBBJYMSMWIIQGU-ZBJDZAJPSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCC=O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
[2H]C([2H])([2H])C([2H])([2H])C=O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C3H6O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
63.11 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
198710-93-9 | |
| Record name | Propionaldehyde-2,2,3,3,3-D5 | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
Synthesis routes and methods I
Procedure details








Synthesis routes and methods II
Procedure details








Synthesis routes and methods III
Procedure details










Synthesis routes and methods IV
Procedure details





Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.

![4-Bromo-6-chlorobenzo[c][1,2,5]thiadiazole](/img/structure/B596181.png)