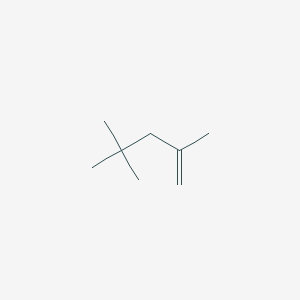
2,4,4-Trimethyl-1-pentene
Overview
Description
2,4,4-Trimethyl-1-pentene (TMP, CAS 107-39-1) is a branched alkene with the molecular formula C₈H₁₆ and a molecular weight of 112.22 g/mol . It is a colorless, flammable liquid with a gasoline-like odor, a boiling point of 100–102°C, and a flash point of -7°C, making it highly volatile . TMP is insoluble in water but miscible with organic solvents like acetone, benzene, and chloroform . Industrially, it is used in polymer synthesis, fuel additives, adhesives, and sealants .
Preparation Methods
2,4,4-Trimethyl-1-pentene is primarily synthesized from isobutylene through an olefinic complex decomposition reaction. This process involves connecting two isobutylene molecules head-to-tail under specific conditions, typically in the presence of a catalyst . Common catalysts include acid and metal catalysts. The reaction is carried out under controlled temperature and pressure to ensure optimal results . The industrial production process includes the following steps:
Raw Material Pretreatment: Purifying and drying isobutylene to meet reaction requirements.
Reaction: Introducing pretreated isobutylene into a reactor to undergo the olefin complex decomposition reaction.
Product Separation and Purification: Separating and purifying the reaction products to obtain high-purity diisobutylene.
Catalyst Regeneration: Regenerating the used catalyst to restore its activity for reuse.
Chemical Reactions Analysis
Hydrocracking for Catalyst Evaluation
TMP1 serves as a model compound for studying hydrocracking (HCG) catalysts. Sulfided CoMo catalysts cleave its C–C bonds under hydrogen pressure to produce lighter hydrocarbons like propane and isobutane . This reaction helps evaluate catalyst acidity and hydrogenation activity.
Reaction Conditions :
-
Temperature: 250–400°C
-
Pressure: Hydrogen-rich environment
Ozonolysis in Flow Reactors
Ozonolysis of TMP1 generates ozonides, which decompose to carbonyl compounds. This reaction is studied under pseudo-first-order conditions to understand oxidative cleavage mechanisms .
Mechanistic Pathway :
-
Ozone addition to the double bond → Ozonide formation.
-
Decomposition → Aldehydes/ketones (e.g., formaldehyde, 2,2-dimethylpropanal) .
Liquid-Phase Oxidation
Oxidation of TMP1 with molecular oxygen yields epoxides and hydrogen peroxide, highlighting its utility in peroxide synthesis .
Key Observations :
Isomerization to 2,4,4-Trimethyl-2-pentene (TMP2)
TMP1 isomerizes to TMP2 under acidic or thermal conditions. Contrary to typical alkene stability trends, equilibrium favors TMP1 due to steric strain in TMP2 .
Table 1: Equilibrium Constants for Isomerization
| Temperature (°C) | Equilibrium Constant (K<sub>eq</sub>) | TMP1:TMP2 Ratio |
|---|---|---|
| 50 | 0.45 | 69:31 |
| 80 | 0.29 | 77:23 |
| 110 | 0.18 | 85:15 |
Thermodynamic Data :
Acid-Catalyzed Dimerization
TMP1 is synthesized via acid-catalyzed dimerization of isobutene or dehydration of tert-butanol. Sulfuric acid (60–98%) or strong acid resins achieve >95% yield under closed conditions .
Optimized Conditions :
Example Synthesis :
text350g tert-butanol + 250g 80% H₂SO₄ → 235g TMP1 (94% yield)[3].
Reaction with Hydroxyl Radicals
In the atmosphere, TMP1 reacts with hydroxyl radicals (- OH) at a rate constant of . This reaction contributes to its environmental degradation.
Scientific Research Applications
Industrial Applications
2,4,4-Trimethyl-1-pentene serves as an essential building block in the production of various chemicals and materials:
- Synthetic Rubber and Plastics : It is widely used in the production of synthetic rubber tackifiers and plasticizers. These additives enhance the performance characteristics of rubber and plastic products by improving flexibility and durability .
- Surfactants : The compound is utilized in the synthesis of surfactants that are crucial for detergents, emulsifiers, and wetting agents. These surfactants help reduce surface tension in liquids, facilitating better mixing and spreading .
- Phenolic Resins : this compound acts as a modifier for phenolic resins, which are used in adhesives, coatings, and molded products due to their excellent thermal stability and mechanical properties .
- Epoxy Resins : It is also employed in the formulation of epoxy resins that are used in coatings and composite materials for their strong adhesive properties and resistance to environmental factors .
- Ultraviolet Absorbers : The compound can be transformed into ultraviolet absorbers that protect materials from degradation caused by UV radiation, thus extending their lifespan .
Environmental Considerations
While this compound has numerous applications, it is also associated with environmental concerns. The compound is highly flammable and poses risks of explosion when mixed with air . Additionally, it is toxic to aquatic organisms and can bioaccumulate in fish . Therefore, proper handling and disposal procedures are critical to mitigate its environmental impact.
Toxicity Studies
Research has been conducted to evaluate the toxicity of this compound:
- A study involving Sprague-Dawley rats indicated that exposure to high doses resulted in increased liver and kidney weights, suggesting potential systemic toxicity at elevated levels .
| Study Type | Dose (mg/kg-day) | Observed Effects |
|---|---|---|
| Short-Term | 0, 100, 300, 1000 | Increased liver weights at high doses |
| Long-Term | Various | No significant pathology observed |
Case Study 1: Production Efficiency
A recent patent describes a method for producing this compound with high purity and yield using tert-butanol or isobutanol as raw materials. The method involves a closed reaction process that significantly increases yield (up to 95%) compared to traditional methods . This advancement highlights the importance of optimizing production processes to enhance efficiency in industrial applications.
Case Study 2: Environmental Impact Assessment
An environmental assessment was conducted to evaluate the effects of this compound on aquatic ecosystems. The findings indicated a need for stringent regulations on its use due to its toxic effects on marine life. Recommendations included improved waste management practices and the development of safer alternatives in industrial applications .
Mechanism of Action
The mechanism of action of diisobutylene involves its reactivity with various chemical agents. It reacts with strong oxidizing agents to form oxidized products and with reducing agents to release hydrogen gas . The molecular targets and pathways involved in these reactions are primarily related to its alkene structure, which allows it to undergo addition and polymerization reactions .
Comparison with Similar Compounds
Structural Isomers: 2,4,4-Trimethyl-2-pentene
TMP is often found as a mixture with its structural isomer 2,4,4-trimethyl-2-pentene (commonly termed di-isobutylene). Key differences include:
The isomer mixture (typically 3:1 ratio of 1-pentene to 2-pentene) is marketed as di-isobutylene, a valuable fuel additive .
Branched Alkenes in the C₇–C₉ Range
TMP belongs to a class of branched alkenes evaluated for fuel applications. Comparisons include:
Key Findings :
- Fuel Performance: TMP-based di-isobutylene outperforms linear alkenes in octane enhancement due to its branched structure, which reduces premature combustion (knocking) .
- Combustion Kinetics : TMP exhibits distinct oxidation pathways compared to iso-octane, with faster radical formation due to its unsaturated structure .
Saturated Analogs: 2,2,3-Trimethylpentane
Compared to saturated hydrocarbons like 2,2,3-trimethylpentane , TMP shows:
Functionalized Derivatives: Methanol-TMP Mixtures
Phase equilibrium studies reveal TMP’s miscibility with alcohols:
- Methanol + TMP: Forms azeotropes at 331 K, impacting distillation processes .
Polymer Chemistry
- TMP’s bulky structure in poly[(maleic anhydride)-alt-TMP] (PMP) segregates polymer chains, reducing intra-chain interactions and photoluminescence efficiency. This contrasts with flexible copolymers like poly[(1-octene)-co-ITA] , which exhibit stronger emissions .
Fuel Additives
- Di-isobutylene (TMP mixture) achieves a RON > 100 , outperforming many linear alkenes. Its high branching improves fuel stability and combustion efficiency .
Table 1: Physical Properties of Selected Compounds
| Compound | Molecular Formula | Boiling Point (°C) | Flash Point (°C) | Solubility in Water |
|---|---|---|---|---|
| This compound | C₈H₁₆ | 100–102 | -7 | Immiscible |
| 2,4,4-Trimethyl-2-pentene | C₈H₁₆ | ~100 | -7 | Immiscible |
| Iso-octane | C₈H₁₈ | 99.3 | 7–10 | Immiscible |
Table 2: Application Comparison
| Compound | Key Applications | Advantages | Limitations |
|---|---|---|---|
| TMP | Fuels, polymers, adhesives | High octane, branching stability | Flammability, aquatic toxicity |
| 2-Methyl-1-propene | Plastics, solvents | High reactivity | High volatility |
| 2,2,3-Trimethylpentane | Solvents, fuels | Stability, low toxicity | Lower octane rating |
Biological Activity
2,4,4-Trimethyl-1-pentene (C8H16) is an organic compound classified as an alkene. It is primarily used in industrial applications, particularly in the production of various chemical intermediates and fuels. Understanding its biological activity is crucial for assessing its environmental impact and potential health effects.
- Molecular Formula : C8H16
- Molecular Weight : 112.21 g/mol
- CAS Number : 107-39-1
- IUPAC Name : 2,4,4-trimethylpent-1-ene
Synthesis and Industrial Use
The synthesis of this compound typically involves the dehydration of tert-butanol or isobutanol using sulfuric acid as a catalyst under controlled conditions. Recent advancements have reported yields exceeding 95% when reactions are conducted in closed systems at elevated temperatures . This efficiency makes it a valuable compound in the production of specialty chemicals and fuels.
Biological Activity Overview
The biological activity of this compound can be categorized into several areas:
1. Toxicological Effects
Studies indicate that this compound can cause irritation to the skin and eyes upon exposure. Ingestion may lead to gastrointestinal irritation . The compound's safety data sheets highlight potential health risks associated with high concentrations.
2. Environmental Impact
As a volatile organic compound (VOC), this compound contributes to air pollution and can participate in photochemical reactions leading to ozone formation. Its presence in gasoline formulations has been noted, where it constitutes approximately 0.011% of industrial average gasoline .
3. Biodegradation Studies
Research has shown that alkenes like this compound can undergo microbial degradation in anaerobic environments. This process is essential for assessing the compound's persistence in the environment and its potential for bioaccumulation.
Case Study 1: Health Effects Assessment
A study conducted on the effects of exposure to various alkenes, including this compound, found that prolonged exposure could lead to respiratory issues and skin sensitization. The study involved occupational exposure assessments among workers in chemical manufacturing settings .
Case Study 2: Environmental Remediation
In a remediation project aimed at reducing VOC levels in groundwater contaminated by industrial activities, this compound was monitored alongside other hydrocarbons. Results indicated that while some compounds were effectively degraded by anaerobic bacteria, the degradation rate of this compound was slower compared to simpler alkenes .
Research Findings
Q & A
Q. Basic: What are the recommended methods for synthesizing and purifying 2,4,4-trimethyl-1-pentene to achieve high purity for research purposes?
This compound is typically synthesized via acid-catalyzed dimerization of isobutylene, which often produces a mixture of isomers, including 2,4,4-trimethyl-2-pentene . To isolate the 1-pentene isomer, fractional distillation under reduced pressure (e.g., 101°C boiling point at atmospheric pressure) is critical, with careful temperature control to minimize thermal degradation . Gas chromatography (GC) coupled with mass spectrometry (MS) is recommended for purity verification, as spectral libraries (e.g., NIST) provide reference fragmentation patterns for isomer differentiation .
Q. Basic: How do the thermodynamic properties of this compound, such as enthalpy of vaporization and boiling point, influence its handling in laboratory settings?
The compound’s low boiling point (101°C) and high volatility (vapor pressure: 10 kPa at 38°C) necessitate storage in sealed, fireproof containers under inert gas to prevent evaporation and combustion . Its enthalpy of vaporization (ΔvapH° = 7.05 kcal/mol) indicates significant energy input is required for phase changes in distillation processes . Researchers must use explosion-proof equipment and avoid electrostatic discharge due to its low flash point (-5°C) and flammability range (0.8–4.8 vol% in air) .
Q. Advanced: What experimental approaches are used to resolve discrepancies between observed combustion behavior and existing kinetic models for this compound?
Discrepancies in combustion kinetics, such as flame speed and ignition delay, are addressed through shock tube experiments and laminar flame studies paired with detailed kinetic models . For example, Sun et al. (2019) identified deviations in predicted intermediate radical concentrations (e.g., CH3 and C2H5) by comparing laser-induced fluorescence (LIF) measurements with CHEMKIN simulations. Adjustments to rate constants for β-scission and H-abstraction reactions in the model improved alignment with experimental data .
Q. Advanced: How does the presence of isomeric impurities like 2,4,4-trimethyl-2-pentene affect the chemical reactivity and analysis of this compound in catalytic studies?
Isomeric impurities alter reaction pathways, particularly in acid-catalyzed systems. For instance, in hydration studies, the 2-pentene isomer exhibits slower reaction rates due to steric hindrance around the double bond. Nuclear magnetic resonance (NMR) analysis (e.g., 13C NMR) can distinguish isomers via chemical shift differences (e.g., δ 115–125 ppm for olefinic carbons) . Researchers should employ high-resolution GC-MS or HPLC with chiral columns to quantify isomer ratios and assess their impact on kinetic outcomes .
Q. Advanced: What methodologies are recommended for assessing the environmental persistence and bioaccumulation potential of this compound in aquatic ecosystems?
The Japanese MITI biodegradation test (OECD 301C) classifies the compound as slowly biodegradable (no degradation observed in 28 days), suggesting persistence in soil and water . Bioaccumulation potential is estimated using the log Kow (4.55) and regression models (e.g., log BCF = 2.80), indicating moderate-to-high bioconcentration in fish . Microcosm studies with 14C-labeled analogs are recommended for tracking degradation metabolites and partitioning behavior in sediment-water systems.
Q. Basic: What spectroscopic techniques are most effective for identifying and characterizing this compound in complex mixtures?
- IR Spectroscopy : Distinct C=C stretching vibrations at ~1650 cm⁻¹ and methyl group absorptions at 1375–1465 cm⁻¹ .
- Mass Spectrometry : Base peak at m/z 57 ([(CH3)2C=CH]+) and molecular ion at m/z 112 (C8H16+) .
- UV/Vis : Weak absorption at ~190 nm due to π→π* transitions, useful for quantifying low concentrations in hydrocarbon matrices .
Q. Advanced: In fuel additive research, how do the combustion characteristics of this compound compare to iso-octane, and what implications does this have for engine performance?
Compared to iso-octane, this compound exhibits higher laminar flame speeds (e.g., 35–40 cm/s vs. 30 cm/s at φ = 1.0) due to faster radical propagation in lean conditions . However, its lower research octane number (RON) increases knocking propensity in turbocharged engines. Engine tests show elevated aldehyde emissions (e.g., formaldehyde), necessitating catalytic converter optimization for compliance with emissions standards .
Q. Advanced: What are the challenges in reconciling theoretical predictions of thermodynamic properties with experimental data for this compound?
Group contribution methods (e.g., Joback) underestimate enthalpy of vaporization by ~5% compared to experimental values (7.05 kcal/mol vs. 7.36 kcal/mol) due to non-ideal intermolecular interactions in branched alkenes . Discrepancies in heat capacity (Cp) calculations arise from incomplete anharmonic corrections in density functional theory (DFT). Hybrid methods combining experimental data (e.g., NIST TRC tables) with ab initio calculations (e.g., CBS-QB3) improve accuracy for property databases .
Properties
IUPAC Name |
2,4,4-trimethylpent-1-ene | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C8H16/c1-7(2)6-8(3,4)5/h1,6H2,2-5H3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
FXNDIJDIPNCZQJ-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(=C)CC(C)(C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C8H16, Array | |
| Record name | DIISOBUTYLENE, ISOMERIC COMPOUNDS | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/3225 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID4026765 | |
| Record name | 2,4,4-Trimethyl-1-pentene | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4026765 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
112.21 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Diisobutylene, isomeric compounds appears as a clear colorless liquid with a petroleum-like odor. Flash point 10 °F. Less dense than water and insoluble in water. Vapors heavier than air. Used in the manufacture of other chemicals., Liquid, Dry Powder; Liquid, Colorless liquid with an odor of gasoline; [HSDB], COLOURLESS LIQUID WITH CHARACTERISTIC ODOUR. | |
| Record name | DIISOBUTYLENE, ISOMERIC COMPOUNDS | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/3225 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | 1-Pentene, 2,4,4-trimethyl- | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | Pentene, 2,4,4-trimethyl- | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | Diisobutylene | |
| Source | Haz-Map, Information on Hazardous Chemicals and Occupational Diseases | |
| URL | https://haz-map.com/Agents/2976 | |
| Description | Haz-Map® is an occupational health database designed for health and safety professionals and for consumers seeking information about the adverse effects of workplace exposures to chemical and biological agents. | |
| Explanation | Copyright (c) 2022 Haz-Map(R). All rights reserved. Unless otherwise indicated, all materials from Haz-Map are copyrighted by Haz-Map(R). No part of these materials, either text or image may be used for any purpose other than for personal use. Therefore, reproduction, modification, storage in a retrieval system or retransmission, in any form or by any means, electronic, mechanical or otherwise, for reasons other than personal use, is strictly prohibited without prior written permission. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
Boiling Point |
214.7 °F at 760 mmHg (USCG, 1999), 101.4 °C, 101 °C | |
| Record name | DIISOBUTYLENE, ISOMERIC COMPOUNDS | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/3225 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1442 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
Flash Point |
35 °F (est.) (USCG, 1999), -5 °C, 23 °F (-5 °C) | |
| Record name | DIISOBUTYLENE, ISOMERIC COMPOUNDS | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/3225 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Diisobutylene | |
| Source | Haz-Map, Information on Hazardous Chemicals and Occupational Diseases | |
| URL | https://haz-map.com/Agents/2976 | |
| Description | Haz-Map® is an occupational health database designed for health and safety professionals and for consumers seeking information about the adverse effects of workplace exposures to chemical and biological agents. | |
| Explanation | Copyright (c) 2022 Haz-Map(R). All rights reserved. Unless otherwise indicated, all materials from Haz-Map are copyrighted by Haz-Map(R). No part of these materials, either text or image may be used for any purpose other than for personal use. Therefore, reproduction, modification, storage in a retrieval system or retransmission, in any form or by any means, electronic, mechanical or otherwise, for reasons other than personal use, is strictly prohibited without prior written permission. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1442 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
Solubility |
Insoluble in water. Soluble in ethyl ether, benzene, carbon tetrachloride, Solubility in water: none | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1442 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
Density |
0.715 at 68 °F (USCG, 1999) - Less dense than water; will float, 0.7150 @ 20 °C/4 °C, Relative density (water = 1): 0.7 | |
| Record name | DIISOBUTYLENE, ISOMERIC COMPOUNDS | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/3225 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1442 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
Vapor Density |
3.8 (AIR= 1), Relative vapor density (air = 1): 3.9 | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1442 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
Vapor Pressure |
82.72 mmHg (USCG, 1999), 44.7 [mmHg], 44.7 mm Hg @ 25 °C, Vapor pressure, kPa at 38 °C: 10 | |
| Record name | DIISOBUTYLENE, ISOMERIC COMPOUNDS | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/3225 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Diisobutylene | |
| Source | Haz-Map, Information on Hazardous Chemicals and Occupational Diseases | |
| URL | https://haz-map.com/Agents/2976 | |
| Description | Haz-Map® is an occupational health database designed for health and safety professionals and for consumers seeking information about the adverse effects of workplace exposures to chemical and biological agents. | |
| Explanation | Copyright (c) 2022 Haz-Map(R). All rights reserved. Unless otherwise indicated, all materials from Haz-Map are copyrighted by Haz-Map(R). No part of these materials, either text or image may be used for any purpose other than for personal use. Therefore, reproduction, modification, storage in a retrieval system or retransmission, in any form or by any means, electronic, mechanical or otherwise, for reasons other than personal use, is strictly prohibited without prior written permission. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1442 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
Color/Form |
Colorless liquid | |
CAS No. |
25167-70-8, 107-39-1 | |
| Record name | DIISOBUTYLENE, ISOMERIC COMPOUNDS | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/3225 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | 2,4,4-Trimethyl-1-pentene | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=107-39-1 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | 2,4,4-Trimethyl-1-pentene | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000107391 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Diisobutylene | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=73942 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Diisobutylene | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=8701 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 1-Pentene, 2,4,4-trimethyl- | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | Pentene, 2,4,4-trimethyl- | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | 2,4,4-Trimethyl-1-pentene | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4026765 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 2,4,4-trimethylpentene | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.042.431 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | 2,4,4-trimethylpent-1-ene | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.003.171 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | DIISOBUTYLENE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/N69L73ADVF | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1442 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
Melting Point |
-136.3 °F (USCG, 1999), -93.5 °C, -93 °C | |
| Record name | DIISOBUTYLENE, ISOMERIC COMPOUNDS | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/3225 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1442 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | 2,4,4-TRIMETHYL-1-PENTENE | |
| Source | ILO-WHO International Chemical Safety Cards (ICSCs) | |
| URL | https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0594 | |
| Description | The International Chemical Safety Cards (ICSCs) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace. | |
| Explanation | Creative Commons CC BY 4.0 | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details











Synthesis routes and methods III
Procedure details





Synthesis routes and methods IV
Procedure details





Synthesis routes and methods V
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.