Emtricitabine
Description
Emtricitabine (FTC) is a nucleoside reverse transcriptase inhibitor (NRTI) approved by the FDA in 2003 for the treatment of HIV-1 infection in combination with other antiretroviral agents . Structurally, it is a cytidine analog characterized by a fluorine substitution at position 5 of the pyrimidine ring and a sulfur atom in the sugar moiety, distinguishing it from lamivudine (3TC) . This compound exhibits dual activity against HIV and hepatitis B virus (HBV) by inhibiting viral reverse transcriptase, thereby terminating DNA chain elongation . Its intracellular half-life (~39 hours) allows once-daily dosing, enhancing patient compliance .
Properties
IUPAC Name |
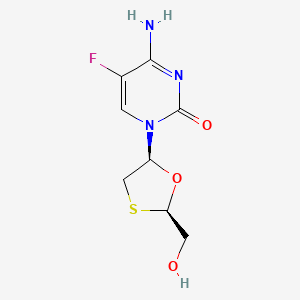
4-amino-5-fluoro-1-[2-(hydroxymethyl)-1,3-oxathiolan-5-yl]pyrimidin-2-one | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C8H10FN3O3S/c9-4-1-12(8(14)11-7(4)10)5-3-16-6(2-13)15-5/h1,5-6,13H,2-3H2,(H2,10,11,14) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
XQSPYNMVSIKCOC-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1C(OC(S1)CO)N2C=C(C(=NC2=O)N)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C8H10FN3O3S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID60861371 | |
| Record name | 4-Amino-5-fluoro-1-[2-(hydroxymethyl)-1,3-oxathiolan-5-yl]pyrimidin-2(1H)-one | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID60861371 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
247.25 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
143491-54-7 | |
| Record name | Racivir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB12753 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Preparation Methods
Early Synthetic Approaches
The foundational synthesis of emtricitabine, as disclosed in U.S. Patent 5,696,254, involves stereoselective reduction of L-menthyl this compound using lithium aluminum hydride (LAH) in tetrahydrofuran (THF). This method achieved an 80% yield but faced criticism for its reliance on hazardous reagents like LAH, which poses handling risks due to its corrosive nature. Additionally, the isolation of this compound required column chromatography, yielding a gummy solid that complicated large-scale production.
Improved Industrial Processes
A patent-pending innovation (WO2014174532A2) addresses these limitations by substituting LAH with sodium borohydride in a phosphate-buffered polar solvent system. This single-step reduction of L-menthyl this compound eliminates intermediate salt isolation, streamlining the process. Key steps include:
- Lot-wise sodium borohydride addition at 5–15°C to minimize side reactions.
- pH-controlled isolation using aqueous acid (pH 5.0–6.5) and base (pH 8.0–8.5) to precipitate impurities.
- Crystallization from isopropanol or acetone to achieve 70% yield with 99.9% purity.
This method enhances safety and scalability while reducing organic solvent use, making it industrially viable (Table 1).
Table 1: Comparative Analysis of this compound Synthesis Methods
Prodrug and Nanoformulation Strategies
ProTide Synthesis for Enhanced Bioavailability
Recent advancements focus on bypassing this compound’s intracellular activation barriers. A 2019 study synthesized a lipophilic prodrug, M2FTC, by reacting this compound with phenylalanine docosyl phosphochloridate in anhydrous THF under argon. The process yielded 61% M2FTC after silica column purification, confirmed via nuclear magnetic resonance (NMR). The prodrug’s design incorporates a docosyl ester to improve nanoparticle stability and monophosphate delivery, critical for long-acting (LA) formulations.
Nanoformulation Techniques
M2FTC was nanoformulated using high-pressure homogenization with poloxamer 407 (P407) as a stabilizer. The resulting nanosuspensions (NM2FTC) exhibited:
- Particle size : 150–200 nm (dynamic light scattering).
- Encapsulation efficiency : 95% (ultra-performance liquid chromatography).
- Stability : 2 months at 37°C without aggregation.
In vitro release studies demonstrated sustained drug release over 30 days, aligning with LA antiretroviral objectives. Transmission electron microscopy revealed spherical morphology, ensuring consistent cellular uptake.
Comparative Analysis of Methods
Traditional chemical methods prioritize yield but face scalability challenges, whereas prodrug nanoformulations prioritize pharmacokinetic optimization. The industrial sodium borohydride method strikes a balance, offering moderate yields with enhanced safety. Nanoformulations, while resource-intensive, extend drug half-life, reducing dosing frequency—a critical advantage in HIV management.
Chemical Reactions Analysis
Types of Reactions: Emtricitabine undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized to form its corresponding sulfoxide and sulfone derivatives.
Reduction: Reduction reactions can convert this compound back to its original form from its oxidized derivatives.
Substitution: Substitution reactions can occur at the fluorine atom or the amino group on the pyrimidine ring.
Common Reagents and Conditions:
Oxidation: Common oxidizing agents include hydrogen peroxide and peracids.
Reduction: Reducing agents such as sodium borohydride and lithium aluminum hydride are used.
Substitution: Substitution reactions often involve nucleophiles such as amines and thiols.
Major Products:
Oxidation: Sulfoxide and sulfone derivatives.
Reduction: this compound.
Substitution: Various substituted derivatives depending on the nucleophile used.
Scientific Research Applications
Emtricitabine has a wide range of scientific research applications, including:
Chemistry: Used as a model compound for studying nucleoside analogues and their chemical properties.
Biology: Employed in research on viral replication and the mechanisms of antiviral agents.
Medicine: Extensively used in clinical research for the treatment and prevention of HIV and HBV infections.
Industry: Utilized in the development of antiretroviral therapies and fixed-dose combination drugs
Mechanism of Action
Emtricitabine exerts its effects by inhibiting the enzyme reverse transcriptase, which is essential for the replication of HIV. It is phosphorylated intracellularly to its active form, this compound 5’-triphosphate, which competes with the natural substrate, deoxycytidine 5’-triphosphate. This results in the termination of the viral DNA chain and inhibition of viral replication .
Comparison with Similar Compounds
Key Data Tables
Table 1: Comparative Pharmacokinetics and Resistance Profiles
| Parameter | Emtricitabine (FTC) | Lamivudine (3TC) | Elvucitabine |
|---|---|---|---|
| Intracellular Half-Life | 39 hours | 18.5 hours | ~100 hours |
| HIV Resistance (M184V/I) | High | High | Moderate |
| HBV Resistance (rtM204V/I) | 13% (2 years) | 14–32% (2 years) | N/A |
| FDA Approval (HIV) | Yes | Yes | No |
Research Findings and Clinical Implications
- Meta-Analysis Outcomes : FTC and 3TC show equivalent efficacy in HIV treatment, but FTC’s longer half-life supports simpler adherence protocols .
- Combination Therapy Superiority : FTC/TDF or FTC/TAF combinations are first-line for HBV-HIV coinfection due to lower resistance rates and synergistic activity .
- Emerging Alternatives : Elvucitabine’s extended half-life and resistance profile warrant further investigation despite discontinued development .
Biological Activity
Emtricitabine (FTC) is a nucleoside reverse transcriptase inhibitor (NRTI) primarily used in the treatment of HIV-1 infection and as part of pre-exposure prophylaxis (PrEP) to prevent HIV transmission. This article explores its biological activity, pharmacodynamics, clinical applications, and safety profile, supported by data tables and relevant case studies.
This compound functions by inhibiting the reverse transcriptase enzyme, which is crucial for the replication of HIV. By mimicking the natural nucleosides, FTC gets incorporated into the viral DNA chain during reverse transcription, leading to premature termination of the DNA strand. This mechanism effectively reduces viral load in patients and helps maintain immune function.
Pharmacokinetics
This compound is rapidly absorbed after oral administration, with peak plasma concentrations occurring approximately 1-2 hours post-dose. It has a half-life of about 10 hours, allowing for once-daily dosing. The drug is primarily eliminated through renal excretion, necessitating dose adjustments in patients with renal impairment.
| Pharmacokinetic Parameter | Value |
|---|---|
| Bioavailability | ~93% |
| Peak Plasma Concentration (Cmax) | 1.5 mg/L |
| Half-Life | 10 hours |
| Renal Clearance | 0.3 L/h/kg |
Antiviral Activity
This compound has demonstrated potent antiviral activity against HIV-1 in vitro and in clinical settings. In a comparative study with other NRTIs, FTC showed superior efficacy in suppressing viral replication.
Case Study: Efficacy in HIV Treatment
A clinical trial involving 660 subjects evaluated FTC's efficacy when combined with other antiretroviral agents. The primary endpoint was maintaining HIV-1 RNA levels below 50 copies/mL at Week 48. Results indicated that FTC maintained virologic suppression effectively across diverse patient populations.
| Study Group | HIV-1 RNA <50 copies/mL | p-value |
|---|---|---|
| FTC + Tenofovir | 89% | <0.001 |
| Control Group | 75% |
Resistance Profile
Resistance to this compound can develop through mutations in the reverse transcriptase gene of HIV. Notably, the M184V/I mutation confers high-level resistance to FTC but retains susceptibility to other NRTIs such as tenofovir.
Safety and Side Effects
While generally well-tolerated, this compound can cause side effects including nausea, diarrhea, and headache. More serious risks include lactic acidosis and hepatotoxicity, particularly in patients with pre-existing liver conditions.
Monitoring Recommendations
Patients on this compound should undergo regular monitoring of renal function due to its renal clearance pathway. The following monitoring schedule is recommended:
| Monitoring Parameter | Frequency |
|---|---|
| Renal Function (Creatinine) | Baseline, then every 3-6 months |
| Liver Function Tests | Baseline and periodically |
Emerging Research: COVID-19 Applications
Recent studies have explored this compound's potential activity against SARS-CoV-2 by inhibiting the viral RNA-dependent RNA polymerase (RdRp). Preliminary findings suggest that FTC may have a role in treating COVID-19, although further investigation is required to establish its efficacy and safety in this context .
Q & A
Q. What validated analytical techniques are recommended for quantifying Emtricitabine in biological matrices, and how are they methodologically implemented?
Answer: The quantification of this compound in biological samples (e.g., plasma, urine) commonly employs high-performance liquid chromatography (HPLC) and voltammetric methods . For HPLC, reverse-phase C18 columns with UV detection at ~280 nm are standard, using mobile phases like acetonitrile-phosphate buffer (pH 3.0) for optimal separation . Voltammetric techniques, such as differential pulse voltammetry, utilize glassy carbon electrodes in phosphate buffer (pH 7.4) for electrochemical oxidation of this compound, achieving detection limits as low as 0.1 µM . Method validation should include specificity (against metabolites like tenofovir), linearity (1–50 µg/mL range), and intra-day precision (<5% RSD) .
Q. What protocols ensure the chemical stability of this compound during experimental handling and storage?
Answer: this compound is hygroscopic and requires storage at 2–8°C in airtight containers under inert gas (e.g., argon) to prevent degradation . For in vitro studies, prepare stock solutions in methanol or deionized water (freely soluble) and avoid prolonged exposure to light. Stability in biological matrices (e.g., plasma) is maintained at -80°C for up to 6 months, with <10% degradation observed .
Q. What are the critical pharmacokinetic parameters to monitor in preclinical studies of this compound, and how are they optimized?
Answer: Key parameters include oral bioavailability (~93%) , plasma half-life (10–20 hours) , and renal clearance (70–80%) . Optimize bioavailability through co-administration with tenofovir disoproxil fumarate (TDF), which enhances intestinal absorption via prodrug activation. For accurate half-life determination, use non-compartmental pharmacokinetic models (e.g., PKSolver software) to analyze plasma concentration-time curves, accounting for inter-patient variability in renal function .
Advanced Research Questions
Q. How can researchers resolve discrepancies in reported pharmacokinetic half-lives of this compound across clinical studies?
Answer: Discrepancies (e.g., 10 h vs. 20 h half-lives) arise from differences in patient demographics (e.g., renal impairment) and analytical models . To reconcile these:
- Conduct compartmental analysis (e.g., two-compartment model) to distinguish distribution and elimination phases.
- Stratify data by glomerular filtration rate (GFR) and adjust for covariates like age using nonlinear mixed-effects modeling (NONMEM) .
- Validate findings with radiolabeled this compound-d₂,¹⁵N to trace metabolite pathways .
Q. What advanced methodologies are employed for impurity profiling during this compound synthesis, and how are critical impurities controlled?
Answer: Impurity profiling requires LC-MS/MS and ¹H/¹³C-NMR to identify structural analogs (e.g., cis/trans cyclic impurities, dimeric byproducts) . Critical impurities include:
- 2-epi-Emtricitabine (CAS 145281-92-1) : Control reaction temperature (<25°C) during stereoselective synthesis to minimize epimerization.
- This compound Disulfide (CAS 145986-26-1) : Use antioxidant buffers (e.g., 0.1% ascorbic acid) in aqueous reactions .
- Quantify impurities at ≤0.15% using USP-grade reference standards and gradient elution (0.1% TFA in acetonitrile/water) .
Q. How can clinical trial designs mitigate bias when evaluating this compound-based PrEP efficacy in high-risk populations?
Answer: The landmark iPrEx trial (NCT00458393) provides a template:
- Randomization : Stratify participants by baseline HIV risk (e.g., condom use frequency) and geographic region.
- Blinding : Use placebo-matched tablets and independent adjudicators for endpoint confirmation .
- Adherence Monitoring : Measure intracellular this compound-triphosphate levels in peripheral blood mononuclear cells (PBMCs) via LC-MS; detectable levels correlate with 44% risk reduction .
- Statistical Adjustments : Apply modified Poisson regression to account for loss to follow-up and non-adherence .
Q. What strategies address low adherence rates in long-term this compound studies, and how are these confounders statistically managed?
Answer:
- Biomarker Validation : Use urine this compound detection (threshold: ≥1 µg/mL) as an objective adherence metric .
- Digital Tools : Implement real-time electronic pill bottles (e.g., Wisepill) with GPS timestamps.
- Analytical Approaches : Apply per-protocol and as-treated analyses with inverse probability weighting to adjust for adherence-related bias .
Q. What methodologies ensure robust detection of this compound resistance mutations in virological failure studies?
Answer:
- Genotypic Assays : Amplify HIV-1 reverse transcriptase regions via RT-PCR and perform Sanger sequencing (detection limit: 20% mutant prevalence).
- Deep Sequencing : Use Illumina MiSeq for low-frequency variants (≥1% prevalence).
- Phenotypic Assays : Measure IC₅₀ shifts in MT-4 cell lines expressing patient-derived RT mutations (e.g., M184V) .
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Featured Recommendations
| Most viewed | ||
|---|---|---|
| Most popular with customers |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.


