Trametinib
Description
Direct Allosteric Inhibition of MEK1/MEK2 Kinase Activity
Trametinib is characterized as an allosteric inhibitor of MEK1 and MEK2. nih.govnih.govprobes-drugs.orgresearchgate.netnih.govresearchgate.net Unlike ATP-competitive inhibitors that bind to the active site, this compound binds to an allosteric site adjacent to the ATP-binding pocket. nih.gov This binding event induces a conformational change in MEK1 and MEK2, locking them in an inactive state and thereby preventing their catalytic activity. nih.govnih.gov this compound preferentially binds to the unphosphorylated forms of MEK1 and MEK2 with high affinity. nih.govnih.gov Studies have reported half maximal inhibitory concentration (IC50) values for this compound against MEK1 and MEK2 in the low nanomolar range, specifically 0.7–0.9 nmol/L. nih.gov
Prevention of MEK Phosphorylation and Activation by Upstream Kinases
Upstream kinases, notably members of the RAF family (such as BRAF and CRAF), are responsible for phosphorylating and activating MEK1 and MEK2. The allosteric binding of this compound to MEK1 and MEK2 prevents this activating phosphorylation. nih.govnih.govnih.govistanbul.edu.tr By maintaining MEK in an unphosphorylated state, this compound effectively blocks the signal transduction from RAF to MEK within the MAPK pathway. nih.gov This inhibition of MEK phosphorylation is crucial for disrupting the constitutively active MAPK signaling often observed in cancers driven by mutations in BRAF or RAS. nih.govprobes-drugs.org
Downstream Impact on ERK1/2 Phosphorylation and Nuclear Translocation
MEK1 and MEK2 are the direct upstream kinases responsible for phosphorylating and activating Extracellular signal-Regulated Kinases 1 and 2 (ERK1 and ERK2). nih.govnih.govnih.govmdpi.com Active MEK1/2 phosphorylates ERK1/2 on specific threonine and tyrosine residues, which is essential for their activation. nih.govscbt.com By inhibiting MEK1/2 kinase activity and preventing their activation, this compound leads to a significant reduction in the phosphorylation of ERK1 and ERK2. nih.govnih.govresearchgate.netistanbul.edu.trscbt.comjuriskes.comwikipedia.orgmdpi.com This decreased phosphorylation of ERK1/2 disrupts their downstream signaling, including their dimerization and subsequent translocation into the nucleus, where they typically phosphorylate and activate various transcription factors involved in cell proliferation, differentiation, and survival. nih.govscbt.com Studies have shown sustained inhibition of ERK phosphorylation in various cancer cell lines and xenograft models treated with this compound. juriskes.comwikipedia.orgmdpi.com
Effects on Cellular Processes in Neoplastic Cells
The inhibition of the MEK/ERK pathway by this compound translates into significant effects on key cellular processes in neoplastic cells, contributing to its anti-tumor activity.
Induction of G1 Cell Cycle Arrest
Constitutive activation of the MAPK pathway often drives uncontrolled cell cycle progression in cancer cells. By inhibiting MEK1/2 and subsequently reducing activated ERK1/2, this compound disrupts the signaling pathways that promote cell cycle progression. kaist.ac.kr This leads to an accumulation of cells in the G1 phase of the cell cycle, effectively inducing a G1 cell cycle arrest. nih.govmdpi.commdpi.comkaist.ac.krresearchgate.netresearchgate.netgenecards.org Research findings support this mechanism, demonstrating increased proportions of cells in the G1 phase after this compound treatment in various cancer cell lines, including melanoma and non-small cell lung cancer cells. mdpi.comresearchgate.net For instance, studies in BRAF-mutant non-small cell lung cancer cells showed that this compound was effective in causing G1 arrest. mdpi.com Similarly, in RAS-/RAF-mutated melanoma cells, this compound induced prolonged G1 arrest. researchgate.net
Promotion of Apoptosis
Inhibition of the pro-survival signals mediated by the constitutively active MAPK pathway can trigger programmed cell death, or apoptosis, in susceptible cancer cells. This compound's blockade of MEK/ERK signaling contributes to the induction of apoptosis in neoplastic cells. nih.govmdpi.commdpi.comgenecards.orgciteab.comfrontiersin.org Studies have shown an increase in markers of apoptosis, such as cleaved caspase-3 and cleaved PARP-1, following this compound treatment in various cancer models. juriskes.comciteab.com For example, in pancreatic cancer xenograft models, this compound treatment was associated with an increase in apoptosis-associated proteins. juriskes.com In vitro studies in sensitive cell lines have also demonstrated a marked increase in cell death, including apoptosis, upon treatment with this compound. citeab.com
Properties
IUPAC Name |
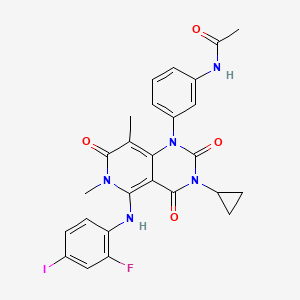
N-[3-[3-cyclopropyl-5-(2-fluoro-4-iodoanilino)-6,8-dimethyl-2,4,7-trioxopyrido[4,3-d]pyrimidin-1-yl]phenyl]acetamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C26H23FIN5O4/c1-13-22-21(23(31(3)24(13)35)30-20-10-7-15(28)11-19(20)27)25(36)33(17-8-9-17)26(37)32(22)18-6-4-5-16(12-18)29-14(2)34/h4-7,10-12,17,30H,8-9H2,1-3H3,(H,29,34) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
LIRYPHYGHXZJBZ-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=C2C(=C(N(C1=O)C)NC3=C(C=C(C=C3)I)F)C(=O)N(C(=O)N2C4=CC=CC(=C4)NC(=O)C)C5CC5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C26H23FIN5O4 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID901007381 | |
| Record name | N-{3-[3-Cyclopropyl-5-(2-fluoro-4-iodoanilino)-6,8-dimethyl-2,4,7-trioxo-3,4,6,7-tetrahydropyrido[4,3-d]pyrimidin-1(2H)-yl]phenyl}ethanimidic acid | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID901007381 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
615.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
871700-17-3 | |
| Record name | Trametinib | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=871700-17-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Trametinib [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0871700173 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Trametinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08911 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | N-{3-[3-Cyclopropyl-5-(2-fluoro-4-iodoanilino)-6,8-dimethyl-2,4,7-trioxo-3,4,6,7-tetrahydropyrido[4,3-d]pyrimidin-1(2H)-yl]phenyl}ethanimidic acid | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID901007381 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | N-{3-[3-cyclopropyl-5-[(2-fluoro-4-iodophenyl)amino]-6,8-dimethyl-2,4,7-trioxo-3,4,6,7-tetrahydropyrido[4,3-d]pyrimidin-1(2H)-yl]phenyl}acetamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | TRAMETINIB | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/33E86K87QN | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Melting Point |
293-303 | |
| Record name | Trametinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08911 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Molecular and Cellular Mechanisms of Action
Effects on Cellular Processes in Neoplastic Cells
Modulation of Cell Survival Pathways
Trametinib, a highly selective, reversible, allosteric inhibitor of MEK1 and MEK2, primarily exerts its effects by blocking the catalytic activity of these kinases within the RAS-RAF-MEK-ERK pathway. This inhibition prevents the phosphorylation and activation of MEK, consequently disrupting downstream signaling crucial for cell proliferation and survival drugbank.comcancercareontario.ca. The MAPK/ERK pathway, frequently activated in cancers due to mutations in genes like RAS and BRAF, plays a vital role in regulating cellular behaviors, including proliferation, invasion, and migration mdpi.comdovepress.com.
Inhibition of MEK by this compound leads to a decrease in phosphorylated ERK (p-ERK) expression, which is associated with reduced cell proliferation spandidos-publications.com. However, the cellular response to MEK inhibition is complex, and cancer cells can employ compensatory mechanisms to maintain survival. One significant pathway involved in cell survival is the PI3K/Akt/mTOR pathway mdpi.comiiarjournals.orgoncotarget.com. Studies have shown that MEK inhibition by this compound can sometimes lead to the activation of the PI3K/Akt pathway, potentially limiting the efficacy of this compound monotherapy mdpi.comnih.gov. This feedback activation of PI3K/Akt can occur through various mechanisms, including the modulation of scaffolding proteins or feedback regulation of receptor tyrosine kinase expression, such as EGFR mdpi.comnih.gov. Hyperactivation of the PI3K/Akt pathway in response to this compound has been observed in various cancer types, including head and neck squamous cell carcinoma (HNSCC) and meningioma mdpi.comnih.gov.
Research has demonstrated that combining this compound with inhibitors targeting the PI3K/Akt/mTOR pathway can enhance anti-tumor effects and overcome resistance mechanisms mdpi.comiiarjournals.orgoncotarget.comnih.gov. For instance, in HNSCC cell lines, combining this compound with PI3K inhibitors like GDC-0941 (pictilisib) or BYL719 (alpelisib) prevented Akt hyperactivation induced by this compound and showed synergistic anti-tumor effects nih.gov. Similarly, studies in melanoma cells with acquired resistance to this compound have explored combinations with metformin, which can suppress mTOR activity, to potentially overcome resistance iiarjournals.orgoncotarget.com.
This compound also modulates apoptotic and anti-apoptotic signaling. While MEK inhibition can induce apoptosis by interfering with ERK-mediated pathways mdpi.comnih.gov, cancer cells can upregulate anti-apoptotic proteins as a survival mechanism. For example, in KRAS-mutant colorectal cancer cells, treatment with low-dose this compound was found to increase the expression of the anti-apoptotic protein Bcl-xL, contributing to drug resistance spandidos-publications.com. Combining this compound with a Bcl-xL antagonist, such as ABT263, demonstrated a synergistic effect on inducing apoptosis in these cells, associated with increased expression of the pro-apoptotic protein BIM and reduced Bcl-xL levels spandidos-publications.comuni-tuebingen.de.
Furthermore, this compound's impact on cell survival pathways extends to the modulation of necroptosis, a form of programmed cell death. Studies, such as those investigating this compound's protective effects against cisplatin-induced acute kidney injury, have shown that this compound can significantly inhibit both apoptosis and necroptosis mdpi.com. This involves reducing the expression of key proteins in the necroptotic pathway, including RIPK1, RIPK3, and p-MLKL mdpi.com.
Detailed research findings highlight the intricate interplay between the MAPK/ERK pathway and other survival pathways, such as PI3K/Akt/mTOR, in determining the response to this compound. The ability of cancer cells to activate compensatory survival mechanisms underscores the rationale for combination therapies targeting multiple pathways to enhance efficacy and overcome resistance.
Below are some data points illustrating the effects of this compound and combination therapies on cell viability and signaling proteins in various cancer cell lines:
| Cell Line | Treatment | Effect on Cell Viability (IC50 or % Inhibition) | Modulation of p-ERK | Modulation of p-AKT | Modulation of Bcl-xL | Modulation of BIM | Reference |
| BON1 (NET) | This compound | IC50: 0.44 nM | Decreased | Not specified | Not specified | Not specified | mdpi.com |
| QGP-1 (NET) | This compound | IC50: 6.359 nM | Decreased | Not specified | Not specified | Not specified | mdpi.com |
| NCI-H727 (NET) | This compound | IC50: 84.12 nM | Decreased | Not specified | Not specified | Not specified | mdpi.com |
| HNSCC Cell Lines | This compound | Reduced cell viability | Decreased | Increased | Not specified | Not specified | nih.gov |
| HNSCC Cell Lines | This compound + PI3K Inhibitor (GDC-0941/BYL719) | Enhanced anti-tumor effect (synergistic) | Decreased | Decreased | Not specified | Not specified | nih.gov |
| KRAS-mutant CRC | Low-dose this compound | Suppressed proliferation | Decreased | Not specified | Increased | Not specified | spandidos-publications.com |
| KRAS-mutant CRC | Low-dose this compound + ABT263 | Synergistic increase in apoptosis | Decreased | Not specified | Reduced | Increased | spandidos-publications.comuni-tuebingen.de |
| Melanoma Cell Lines | Metformin + this compound + Paclitaxel | Differential growth inhibition (synergistic/antagonistic) | Variable (cell line dependent) | Affected mTOR pathway | Not specified | Not specified | iiarjournals.org |
| Ovarian Cancer Cells | This compound + JQ1 (BET inhibitor) | Synergistic increase in apoptosis | Not specified | Reduced p-BAD | Not specified | Increased | oncotarget.com |
Interactive Table 1: Effects of this compound and Combination Therapies on Cell Viability and Signaling Proteins
| Cell Line | Treatment | Effect on Cell Viability (IC50 or % Inhibition) | Modulation of p-ERK | Modulation of p-AKT | Modulation of Bcl-xL | Modulation of BIM | Reference |
| BON1 (NET) | This compound | IC50: 0.44 nM | Decreased | Not specified | Not specified | Not specified | mdpi.com |
| QGP-1 (NET) | This compound | IC50: 6.359 nM | Decreased | Not specified | Not specified | Not specified | mdpi.com |
| NCI-H727 (NET) | This compound | IC50: 84.12 nM | Decreased | Not specified | Not specified | Not specified | mdpi.com |
| HNSCC Cell Lines | This compound | Reduced cell viability | Decreased | Increased | Not specified | Not specified | nih.gov |
| HNSCC Cell Lines | This compound + PI3K Inhibitor (GDC-0941/BYL719) | Enhanced anti-tumor effect (synergistic) | Decreased | Decreased | Not specified | Not specified | nih.gov |
| KRAS-mutant CRC | Low-dose this compound | Suppressed proliferation | Decreased | Not specified | Increased | Not specified | spandidos-publications.com |
| KRAS-mutant CRC | Low-dose this compound + ABT263 | Synergistic increase in apoptosis | Decreased | Not specified | Reduced | Increased | spandidos-publications.comuni-tuebingen.de |
| Melanoma Cell Lines | Metformin + this compound + Paclitaxel | Differential growth inhibition (synergistic/antagonistic) | Variable (cell line dependent) | Affected mTOR pathway | Not specified | Not specified | iiarjournals.org |
| Ovarian Cancer Cells | This compound + JQ1 (BET inhibitor) | Synergistic increase in apoptosis | Not specified | Reduced p-BAD | Not specified | Increased | oncotarget.com |
Interactive Table 2: Effects of this compound on Necroptosis Markers in Cisplatin-Injected Mice Kidneys
| Protein | Control (Relative Expression) | Cisplatin (Relative Expression) | Cisplatin + this compound (Relative Expression) | p-value (Cisplatin vs. Cisplatin + this compound) | Reference |
| RIPK1 | Not specified | 10.51 ± 0.39 | 1.78 ± 0.18 | < 0.001 | mdpi.com |
| RIPK3 | Not specified | 12.82 ± 0.63 | 2.08 ± 0.28 | < 0.001 | mdpi.com |
| p-MLKL | Not specified | 2.62 ± 0.31 | 0.84 ± 0.06 | < 0.01 | mdpi.com |
These findings underscore the multifaceted impact of this compound on cell survival pathways, highlighting its primary role in inhibiting the MAPK/ERK pathway while also revealing the complexity introduced by feedback loops and compensatory mechanisms involving pathways like PI3K/Akt/mTOR and the modulation of apoptotic and necroptotic signaling.
Preclinical Investigation of Trametinib Monotherapy
In Vivo Efficacy Studies
Suppression of Tumor Growth in BRAF V600 Mutation-Positive Tumor Xenografts
Preclinical data have demonstrated the efficacy of trametinib in inhibiting tumor growth in xenograft models harboring the BRAF V600E mutation. In a BRAFV600E-containing xenograft model of human melanoma (A375P), orally administered dabrafenib, a BRAF inhibitor, inhibited ERK activation and led to tumor growth inhibition. plos.org While this specific study focused on dabrafenib monotherapy and combination, other research highlights this compound's potent activity against BRAF-mutant models. For instance, in 8505C tumor xenografts bearing a BRAFV600E mutation, this compound alone was highly effective, resulting in sustained tumor size reductions. spandidos-publications.com Similarly, a patient-derived orthotopic xenograft (PDOX) model of BRAF-V600E-mutant melanoma showed that this compound significantly regressed tumor growth compared to the untreated control. researchgate.net This regression was observed from day 3 to day 14, with this compound demonstrating significantly greater efficacy than other tested therapies, including vemurafenib, cobimetinib, and temozolomide, by day 7. researchgate.net
Sustained Inhibition of ERK Phosphorylation in Xenograft Models
This compound's mechanism of action involves the inhibition of MEK1 and MEK2, which are directly upstream of ERK in the MAPK pathway. mims.com Preclinical studies consistently show that this compound treatment leads to the inhibition of ERK phosphorylation in xenograft models. In human tumor xenograft models, this compound induced rapid and sustained dephosphorylation of phosphorylated MEK and, consequently, inhibited phosphorylated ERK. nih.gov This inhibition of ERK phosphorylation is considered a key biomarker of MEK inhibitor activity. nih.gov For example, in pancreatic ductal adenocarcinoma (PDAC) xenografts, this compound treatment specifically decreased phosphorylated ERK levels. nih.gov Similarly, immunohistochemical analysis of thyroid cancer xenografts showed that ERK activation was significantly inhibited by this compound. spandidos-publications.com In a mouse xenograft model of malignant pleural mesothelioma, this compound blocked MEK-dependent ERK phosphorylation in a concentration-dependent manner. nih.gov Tumors from mice treated with this compound also showed the weakest staining for p-ERK. nih.gov
Impact on Ki67 Expression in Tumor Xenografts
Ki67 is a nuclear protein associated with cellular proliferation. Preclinical investigations have examined the effect of this compound on Ki67 expression in tumor xenograft models as an indicator of reduced cell division. In a BRAFV600E-containing xenograft model, dabrafenib, a BRAF inhibitor often used in combination with this compound, downregulated Ki67 expression, contributing to tumor growth inhibition. plos.org While direct data on this compound monotherapy's impact on Ki67 in BRAF V600E xenografts was less prominent in the immediate search results, studies in other xenograft models provide insight. In PDAC xenografts, effects of therapy on intratumoral proliferation corresponded with tumor growth inhibition, and this compound's effects were accompanied by changes in markers including those associated with proliferation. nih.gov Immunohistochemical staining of xenografts in an intrahepatic cholangiocarcinoma model revealed that Ki-67 staining was weakest in the this compound treatment group, indicating reduced proliferation. nih.gov Studies using different cancer cell lines in xenograft models also showed that this compound treatment led to a decrease in Ki67 expression, with a strong dose-dependent decrease observed in some sensitive cell lines. researchgate.net
Therapeutic Potential in Noonan Syndrome Preclinical Models for Cardiovascular Phenotype Amelioration
Noonan Syndrome is a developmental disorder often associated with mutations in the RAS/MAPK pathway, leading to various clinical features, including cardiovascular defects like hypertrophic cardiomyopathy. thrasherresearch.orgjacc.org Preclinical studies in mouse models of Noonan Syndrome have investigated the potential of MEK inhibitors like this compound to ameliorate these cardiovascular phenotypes. Research using a preclinical mouse model of Noonan Syndrome carrying the germline mutation RIT1M90I, which exhibit classic features including cardiovascular defects, assessed the efficacy of oral this compound. thrasherresearch.org Results indicated that daily oral administration of this compound significantly improved the cardiovascular phenotype of these mice. thrasherresearch.org A statistically significant decrease in heart weight-to-body weight ratio was observed in this compound-treated mice compared to placebo littermates. thrasherresearch.org Additionally, the studies showed a decrease in proliferating cells in the heart tissue of treated mice. thrasherresearch.org These findings suggest that MEK inhibition with this compound holds therapeutic potential for addressing the cardiovascular manifestations of Noonan Syndrome in preclinical settings. nih.govjacc.org
Efficacy in Thyroid Cancer Xenografts (BRAF- and RAS-Mutant Models)
Thyroid cancer frequently exhibits activation of the ERK pathway due to mutations in genes such as BRAF and RAS. spandidos-publications.com Preclinical studies have evaluated the efficacy of this compound in thyroid cancer xenograft models harboring these mutations. This compound has shown marked single-agent activity against both RAS- and BRAF-mutant thyroid cancer models in vitro and in vivo. spandidos-publications.com In xenografts bearing either KRASG12R or BRAFV600E mutations, this compound alone was highly effective, leading to sustained shrinkage in tumor volume compared to baseline. spandidos-publications.com Specifically, this compound was effective as a single agent against the RAS mutant CAL62 xenograft model. spandidos-publications.com These preclinical findings support the evaluation of this compound as a potential therapeutic agent in thyroid cancer with BRAF and RAS mutations. spandidos-publications.commdpi.com
Preclinical Study Findings Summary
| Preclinical Model / Finding | Key Observation(s) | Relevant Section | Source(s) |
| BRAF V600E Melanoma Xenografts | Sustained tumor size reductions with this compound monotherapy; significant tumor regression compared to control. | 3.2.1 | spandidos-publications.comresearchgate.net |
| Various Tumor Xenografts (Melanoma, PDAC, Thyroid, Mesothelioma) | Rapid and sustained inhibition of ERK phosphorylation; decreased phosphorylated ERK levels in tumor tissue. | 3.2.2 | nih.govspandidos-publications.comnih.govnih.govnih.gov |
| Various Tumor Xenografts (Melanoma, PDAC, ICC) | Downregulation or decrease in Ki67 expression, correlating with reduced cell proliferation. | 3.2.3 | nih.govplos.orgnih.govresearchgate.net |
| Noonan Syndrome Mouse Model (RIT1M90I mutation) | Significant improvement in cardiovascular phenotype; decreased heart weight-to-body weight ratio; reduced proliferating cells in the heart. | 3.2.4 | nih.govthrasherresearch.orgjacc.org |
| Thyroid Cancer Xenografts (BRAF- and RAS-mutant) | Marked single-agent activity against both BRAF- and RAS-mutant models; sustained tumor shrinkage. | 3.2.5 | spandidos-publications.com |
Mechanisms of Acquired and Intrinsic Resistance to Trametinib
Reactivation of the MAPK Pathway
Reactivation of the MAPK pathway is a primary mechanism of both acquired and intrinsic resistance to trametinib. mdpi.comnih.gov This can occur through alterations at different levels of the pathway, bypassing the MEK inhibition. mdpi.com
Activation of receptor tyrosine kinases (RTKs), such as EGFR, can lead to the reactivation of the MAPK pathway and confer resistance to MEK inhibitors like this compound. spandidos-publications.comtandfonline.comnih.gov Studies have shown that this compound treatment can lead to the activation of EGFR and HER2 signals in some cancer cells, reactivating ERK1/2 phosphorylation which was inhibited by this compound. spandidos-publications.com This suggests that EGFR and HER2 activation can serve as a bypass mechanism. spandidos-publications.com Combining this compound with inhibitors targeting EGFR and HER2 has shown synergistic effects in overcoming this resistance in preclinical models. spandidos-publications.comnih.gov
Alterations in the BRAF gene are frequently observed resistance mechanisms, particularly in BRAF-mutant melanomas treated with BRAF and MEK inhibitors. mdpi.comfrontiersin.orgresearchgate.net These alterations can include BRAF splicing variations and amplification. mdpi.comfrontiersin.orgresearchgate.net For instance, a novel BRAF splicing isoform lacking exons 2-10 has been detected in patients resistant to combined dabrafenib and this compound therapy. mdpi.comresearchgate.netaacrjournals.org BRAF amplification has also been associated with resistance in melanoma and colon cancer. mdpi.comfrontiersin.orgresearchgate.net These alterations can lead to the expression of truncated or overexpressed BRAF proteins that constitutively activate the downstream MAPK pathway, bypassing MEK inhibition. researchgate.netoncotarget.com
Mutations in the genes encoding MEK1 and MEK2 (MAP2K1 and MAP2K2) are direct mechanisms of resistance to this compound, as they are the direct targets of the drug. nih.govmdpi.comaacrjournals.orgsoton.ac.uk These mutations can alter the drug binding site or enhance the intrinsic kinase activity of MEK1/2, rendering them less sensitive to this compound inhibition. nih.gov MEK1 mutations have been detected in this compound-resistant cells and patient tumors. nih.govmdpi.com While MEK1 and MEK2 share high sequence homology, functional differences exist, and specific mutations like MEK2C125S have been shown to confer resistance to combination therapy. mdpi.comnih.gov
While MEK1/2 are considered the primary activators of ERK1/2, mutations in ERK1 or ERK2 that lead to constitutive activation could theoretically bypass MEK inhibition by this compound. nih.govmdpi.com However, constitutively active ERK mutations have been rarely reported as a mechanism of resistance to this compound or combined BRAF/MEK inhibition. mdpi.com This may be partly due to the unique regulatory mechanisms of ERK activation, where autophosphorylation is the primary means of autonomous activity increase, requiring synergistic mutations. mdpi.com
Beyond the canonical RAS-RAF-MEK-ERK cascade, alternative or non-canonical mechanisms can also lead to ERK1/2 activation and contribute to this compound resistance. mdpi.comnih.govresearchgate.net One such mechanism involves the protein kinase R-like endoplasmic reticulum kinase (PERK). mdpi.comnih.govresearchgate.net Studies have shown that in some contexts, particularly under stress conditions like those induced by targeted therapy, PERK can phosphorylate ERK1/2 upon its translocation to the endoplasmic reticulum. mdpi.comnih.govresearchgate.net This PERK-mediated ERK1/2 activation can promote cytoprotective autophagy, contributing to resistance to BRAF and MEK inhibition. mdpi.comnih.govresearchgate.net
Constitutive ERK Mutation as a Bypass Mechanism
Activation of Alternative Signaling Pathways
In addition to MAPK pathway reactivation, resistance to this compound can also arise from the activation of alternative signaling pathways that can bypass the need for MEK-ERK signaling for cell survival and proliferation. mdpi.comnih.govnih.govnih.gov The PI3K-AKT pathway is a prominent example of an alternative pathway frequently implicated in this compound resistance. nih.govnih.govoncotarget.combiorxiv.org Activation of the PI3K-AKT pathway can occur through various mechanisms, including mutations in PIK3CA, loss of the tumor suppressor PTEN, or activation of upstream RTKs. nih.govoncotarget.combiorxiv.orgpnas.org This pathway can promote cell survival and growth independently of MEK-ERK signaling, thereby conferring resistance to this compound. nih.govoncotarget.combiorxiv.org Other alternative pathways that have been implicated in resistance include the JAK-STAT pathway and the Hippo pathway effector YAP1. nih.govmedrxiv.orgnih.gov Activation of these pathways can provide alternative survival signals or promote a more resistant phenotype. nih.govnih.gov
Here is a summary of some reported resistance mechanisms and associated findings:
Phosphatidylinositide 3-kinase (PI3K)/AKT Pathway Upregulation
Upregulation or activation of the PI3K/AKT pathway is a significant mechanism of both acquired and intrinsic resistance to this compound. aacrjournals.orgdovepress.com This pathway acts as a bypass mechanism, allowing cancer cells to circumvent the MEK blockade and continue to proliferate and survive. Studies have shown increased PI3K/AKT pathway activation in this compound-resistant tumors. aacrjournals.orgdovepress.com This activation can occur through various mechanisms, including genetic alterations in PI3K or AKT genes or activation by upstream signaling molecules. dovepress.com The PI3K/AKT/mTOR pathway is frequently dysregulated in cancers, contributing to uncontrolled cell growth and resistance to apoptosis. vulcanchem.com Inhibiting the PI3K/AKT pathway in combination with MEK inhibitors has been proposed as a strategy to overcome resistance. dovepress.com Research in canine mucosal melanoma, which shares parallels with human mucosal melanoma, demonstrated that resistance to this compound was reversed by inhibiting the activation of the PI3K/AKT/mTOR pathway. biorxiv.org
Role of c-Kit Activation
Activation of c-Kit, a receptor tyrosine kinase, has been implicated in mechanisms of resistance to targeted therapies, including MEK inhibitors. aacrjournals.orgmdpi.com c-Kit signaling can drive proliferation and survival through the activation of downstream pathways such as PI3K and MAPK. mdpi.com While c-KIT mutations are found in a subset of melanomas, including mucosal and acral subtypes, their role in conferring resistance to this compound specifically, independent of other resistance mechanisms, is an area of ongoing research. biorxiv.orgmdpi.com Studies have suggested increased c-Kit activation in resistant tumors. aacrjournals.orgaacrjournals.org
Adaptive Cellular Reprogramming and Phenotype Switching
Adaptive cellular reprogramming and phenotype switching represent dynamic, non-genomic mechanisms by which cancer cells can develop resistance to this compound. This involves rapid changes in gene expression and cellular state. oncoscience.us
Genomic Adaptive Reprogramming and Enhancer Formation (e.g., BRD4 binding)
Adaptive resistance to this compound can involve significant remodeling of the genomic landscape, including the formation of new enhancers. oncoscience.usunclineberger.orgunc.edunih.gov These enhancers are DNA sequences that can increase the transcription of specific genes, allowing cancer cells to adapt and survive in the presence of the drug. unclineberger.orgunc.edu Bromodomain-containing protein 4 (BRD4), a member of the BET family of bromodomain proteins, plays a crucial role in the formation and function of these enhancers. oncoscience.usnih.govoaepublish.com BRD4 binds to acetylated histones and transcription factors, regulating transcriptional elongation. oaepublish.com Upon this compound treatment, rapid and widespread formation of enhancers with high BRD4 density has been observed, particularly near receptor tyrosine kinase (RTK) loci, contributing to their upregulation and subsequent resistance. oncoscience.usnih.govoaepublish.com Inhibiting BRD4 has shown promise in attenuating this adaptive reprogramming and restoring sensitivity to this compound in preclinical models. unclineberger.orgunc.edunih.govoaepublish.com
Epithelial-Mesenchymal Transition (EMT) and Stem-Like Phenotype Adaptation
Epithelial-Mesenchymal Transition (EMT) is a process by which epithelial cells acquire mesenchymal characteristics, often associated with increased invasiveness, migration, and resistance to therapy. Adaptation towards a stem-like phenotype has also been linked to drug resistance. Research suggests that changes in epithelial gene expression and EMT-related signatures can occur with the development of MEK inhibitor resistance. aacrjournals.org Furthermore, some studies indicate a potential shift from a stem-like subtype to an inflammatory subtype in this compound-resistant cells. nih.gov
Inflammatory Gene Expression and NFκB Activation
Acquired resistance to this compound has been associated with the upregulation of inflammatory gene expression and activation of the NFκB signaling pathway. aacrjournals.orgresearchgate.net Studies in colorectal cancer cell lines have shown that this compound resistance is linked to an enrichment of inflammatory gene signatures, with TNFα and NFκB gene sets being highly ranked. researchgate.net NFκB activation can be greater in cell lines resistant to MEK inhibition. aacrjournals.org Inflammatory cytokines or conditioned medium from macrophage cultures can induce resistance to MEK inhibition, and this is associated with increased NFκB activation. aacrjournals.org The bromodomain inhibitor JQ1 has been shown to suppress inflammatory gene expression and NFκB activation, and in combination with MEK inhibitors, it can overcome resistance. aacrjournals.orgnih.gov
Strategies to Overcome Resistance
Addressing this compound resistance involves strategies aimed at counteracting the mechanisms by which cancer cells evade MEK inhibition.
Combination Therapy Approaches
Combining this compound with other targeted agents or therapies is a widely explored strategy to overcome or delay resistance. The rationale is to simultaneously block multiple pathways that contribute to tumor growth and survival or to target the specific resistance mechanisms that emerge.
A prominent example is the combination of this compound with a BRAF inhibitor, such as dabrafenib. This combination has shown significantly improved response rates and progression-free survival compared to BRAF inhibition alone in patients with BRAF V600-mutated melanoma and other solid tumors. ascopubs.orgoncotarget.comfda.govcancer.govnih.govjhoponline.com The combination mitigates a key resistance pathway that involves MEK activation compensating for BRAF inhibition. cancer.govbmj.com
Combination therapies can target various resistance mechanisms:
Targeting the PI3K/AKT pathway: Combining MEK inhibitors with inhibitors of the PI3K/AKT pathway may overcome resistance driven by the activation of this bypass route. aacrjournals.orgnih.gov For instance, studies suggest that combining this compound with a PI3K inhibitor like GSK2126458 can be effective in colorectal cancer cells resistant to this compound. nih.gov
Targeting RTKs: Inhibiting activated RTKs that contribute to resistance, such as MET or EGFR, in combination with this compound, can be a strategy. ascopubs.orgamegroups.orgmdpi.comfrontiersin.org Adding EGFR monoclonal antibodies to BRAF/MEK combinations has shown benefit in colorectal cancer with intrinsic resistance. ascopubs.org Triple-targeted therapy involving EGFR, BRAF, and MEK inhibitors is being investigated for specific resistance scenarios in NSCLC. amegroups.org
Targeting other kinases: Combining this compound with inhibitors of other kinases involved in resistance, such as ERK or BRD4, is under investigation. mdpi.comunclineberger.org Preclinical studies have shown that pairing this compound with an investigational anti-BRD4 drug can prevent the onset of resistance in triple-negative breast cancer models by inhibiting adaptive reprogramming. unclineberger.org
Targeting RAF dimers: Type II RAF inhibitors, which are effective against RAF dimers, in combination with this compound, can overcome resistance to type I RAF and MEK inhibitors, particularly in anaplastic thyroid cancer. medrxiv.org
Data from clinical trials highlight the benefits of combination therapy. The combination of dabrafenib and this compound has demonstrated consistent response rates in various tumor types harboring the BRAF V600E mutation. ascopubs.org
| Tumor Type | Overall Response Rate (ORR) with Dabrafenib + this compound | Citation |
| Biliary tract cancer | 46% | fda.govcancer.gov |
| High-grade gliomas | 33% | fda.govcancer.gov |
| Low-grade gliomas | 50% | cancer.gov |
| Various solid tumors | 41% (across 24 tumor types) | fda.gov |
Intermittent Dosing and Drug Rechallenge Strategies
Intermittent dosing and drug rechallenge are alternative strategies explored to manage or overcome this compound resistance, particularly acquired resistance. The concept behind these approaches is based on the observation that some resistance mechanisms may be reversible or that temporary drug withdrawal could re-sensitize tumor cells to treatment. mdpi.comresearchgate.netnih.govfrontiersin.org
Preclinical research in animal models initially suggested that intermittent dosing of BRAF and MEK inhibitors might delay acquired resistance by altering the selective pressure on tumor cells. onclive.comoncology-central.comecancer.org This hypothesis proposed that intermittent exposure could select for tumor cells that remain sensitive to the treatment for a longer duration. onclive.com
However, clinical trial data have provided contrasting results. The phase 2 SWOG S1320 trial investigated intermittent versus continuous dosing of dabrafenib and this compound in patients with advanced BRAF V600-mutant melanoma. onclive.comoncology-central.comonclive.com The study found that continuous dosing resulted in superior progression-free survival compared to intermittent dosing. onclive.comecancer.orgonclive.com This outcome was contrary to the preclinical findings. onclive.comonclive.com Possible explanations for this discrepancy include differences in drug metabolism and resistance mechanisms between humans and mice, and the potential for non-MAPK resistance pathways to be more dominant in human patients. frontiersin.orgonclive.com
Despite the findings from the SWOG S1320 trial regarding intermittent dosing to delay resistance, drug rechallenge after a period of drug holiday following acquired resistance remains a potential therapeutic option in the salvage setting for some patients. mdpi.comfrontiersin.org It has been demonstrated in preclinical models that certain resistance mechanisms are reversible, and after a "drug holiday," some patients may regain sensitivity. frontiersin.org Studies in this compound-resistant melanoma cell lines have shown that alternating periods of drug withdrawal and rechallenge can be highly responsive, particularly in cells displaying a differentiation phenotype. mdpi.comnih.govdntb.gov.ua This suggests that melanoma cell plasticity plays a role, where resistant cells adapt to altered conditions but can be influenced by drug re-exposure. researchgate.netnih.gov
In clinical practice, rechallenge with dabrafenib and this compound has been explored in patients with metastatic melanoma who progressed after initial targeted therapy and subsequent immunotherapy. frontiersin.org While the duration of the drug holiday did not appear to be associated with the incidence of response in one analysis, the strategy has shown some clinical activity. frontiersin.org
Clinical Development and Therapeutic Applications of Trametinib
Clinical Efficacy as Monotherapy
Trametinib as a single agent has demonstrated clinical activity in patients with BRAF V600-mutated melanoma. However, its efficacy, particularly in terms of progression-free survival and objective response rates, has been reported to be lower compared to BRAF inhibitors used as monotherapy. dovepress.com
Combination Therapy Regimens
The understanding of resistance mechanisms to single-agent BRAF or MEK inhibitors led to the investigation of combination therapy approaches to achieve more durable responses and overcome or delay the emergence of resistance.
Dabrafenib (BRAF Inhibitor) and this compound Combination
The combination of dabrafenib, a BRAF inhibitor, and this compound has become a standard of care for patients with unresectable or metastatic melanoma harboring BRAF V600E or V600K mutations. pensoft.netbecarispublishing.comascopubs.org This combination targets two different kinases within the same signaling pathway. pensoft.net
Results from the COMBI-v trial showed a median progression-free survival of 12.6 months for the combination group compared with 7.3 months for vemurafenib alone. nice.org.uk The objective response rate was 64% for combination therapy and 51% for vemurafenib monotherapy in this trial. pensoft.net A meta-analysis indicated that dabrafenib + this compound significantly prolongs survival outcomes compared with monotherapies. becarispublishing.com
The combination of BRAF and MEK inhibitors has been shown to delay the emergence of resistance compared with BRAF inhibitor monotherapy. researchgate.netascopubs.orgaacrjournals.org This is attributed to the dual blockade of the MAPK pathway, which can prevent or delay resistance mechanisms that arise through pathway reactivation. becarispublishing.com
Here is a data table summarizing key efficacy outcomes from clinical trials comparing dabrafenib and this compound combination therapy to monotherapy in BRAF V600-mutated melanoma:
| Study | Regimen | Median PFS (months) | Median OS (months) | Objective Response Rate (%) |
| COMBI-d | Dabrafenib + this compound | 11.0 nice.org.uk | Not explicitly stated as median in snippet, but OS improved nice.org.uk | Not explicitly stated in snippet |
| COMBI-d | Dabrafenib Monotherapy | 8.8 nice.org.uk | Not explicitly stated in snippet, but OS lower than combo nice.org.uk | Not explicitly stated in snippet |
| COMBI-v | Dabrafenib + this compound | 12.6 nice.org.uk | Not explicitly stated as median in snippet, but OS improved pensoft.netnice.org.uk | 64 pensoft.net |
| COMBI-v | Vemurafenib Monotherapy | 7.3 pensoft.netnice.org.uk | Not explicitly stated in snippet, but OS lower than combo pensoft.netnice.org.uk | 51 pensoft.net |
| Phase I/II | Dabrafenib + this compound (2mg this compound) | Not explicitly stated in snippet | Not explicitly stated in snippet | 76 dovepress.com |
| Phase I/II | Dabrafenib Monotherapy | Not explicitly stated in snippet | Not explicitly stated in snippet | 54 dovepress.com |
While combination therapy can lead to higher rates of adverse events, the toxicity profile of the dabrafenib and this compound combination has been reported as manageable, and in some cases, the combination may even prevent complications associated with BRAF inhibitor monotherapy. ascopubs.orgascopubs.org For example, the combination regimen using full doses of both drugs generated fewer adverse skin events than BRAF inhibitor monotherapy. dovepress.com The lower incidence of squamoproliferative lesions during combined inhibition is possibly related to blocking of paradoxical MAPK signaling downstream of CRAF by MEK inhibition. dovepress.com
Extensive clinical experience with the combination in metastatic melanoma has provided a wealth of strategies to identify and manage associated adverse events. nih.gov Understanding the adverse event profile and possible strategies to mitigate these toxicities is important. ascopubs.orgascopubs.org
Improved Efficacy and Delay of Resistance
Combination with Immunotherapies (e.g., Anti-PD-1, Anti-CTLA-4 Antibodies)
Combining MAPK pathway inhibitors, such as dabrafenib and this compound, with immunomodulatory antibodies targeting CTLA-4 or PD-L1/PD-1 is an area of significant clinical interest. aacrjournals.org Preclinical studies have explored the potential synergistic effects of these combinations. oncotarget.comnih.gov
In preclinical models, the addition of an anti-PD-1 antibody to combined dabrafenib and this compound was associated with superior outcomes compared with relevant controls. targetedonc.com Preclinical evidence has also shown that combined therapy of dabrafenib, this compound, and anti-PD-1 provided superior antitumor activity against established BRAF V600E mutant murine melanoma compared with anti-PD-1 plus either therapy alone, or isotype control with both dabrafenib and this compound. nih.gov In vitro studies indicated that the combination of dabrafenib, this compound, and anti-PD-1 increases CD8+ tumor infiltrating lymphocytes (TILs), as well as CD4+ T cells and tumor-associated macrophages (TAMs). nih.gov
Clinical trials are evaluating the efficacy and tolerability of combining targeted therapy (dabrafenib and this compound) with immunotherapy (e.g., pembrolizumab). aacrjournals.org While preclinical data support the synergistic effect of both modalities, data confirming the activity and tolerability of these combinations are still being gathered in the clinical setting. oncotarget.com
Combination with PI3K/AKT/mTOR Inhibitors (e.g., Everolimus, GSK2141795)
The combination of MEK inhibitors like this compound with inhibitors of the PI3K/AKT/mTOR pathway has been investigated as a strategy to overcome resistance mechanisms and enhance therapeutic efficacy in various cancer types. Preclinical studies have shown that therapies are more effective when drugs targeting the PI3K-Akt-mTOR pathway are combined with inhibitors of other pathways, such as RAS-MEK-ERK. mdpi.com
A phase I trial investigated the safety, tolerability, and recommended phase 2 dose for the pan-PI3K/mTOR inhibitor, GSK2126458, and this compound combination when administered to patients with advanced solid tumors. nih.gov While mutations in RAS/RAF/PI3K were detected in a significant percentage of patients, minimal responses were observed, potentially due to overlapping toxicities that limited sufficient dose exposure. nih.gov
The combination of this compound and the PI3K/mTOR dual inhibitor GSK2126458 also enhanced cell growth inhibition in B-Raf inhibitor-resistant BRAF mutant melanoma cell lines. oncotarget.com
A phase I trial is studying the side effects and best dose of neratinib in combination with everolimus, palbociclib, or this compound in participants with solid tumors with specific mutations (EGFR, HER2, HER3/4, or KRAS) that are refractory and metastatic. mdanderson.org Giving neratinib with everolimus, palbociclib, or this compound may work better than neratinib alone in treating these participants. mdanderson.org
A phase I trial evaluated the combination of this compound and everolimus in pediatric and young adult patients with recurrent low-grade and high-grade gliomas. cancer.govucsf.edu This study aims to determine the recommended phase 2 dose of this combination. ucsf.edu
A single-arm, single-stage phase II trial is evaluating the combination of this compound (GSK1120212) and GSK2141795, an AKT inhibitor, in patients with recurrent or persistent cervical cancer. dana-farber.org The study aims to determine if this combination is useful in treating this type of cancer and if tumors with a particular genetic makeup show a better response. dana-farber.org Another phase II trial studied how well this compound and Akt inhibitor GSK2141795 work in treating patients with metastatic triple-negative breast cancer. mycancergenome.org This trial included a design where patients initially received this compound monotherapy and, upon disease progression, continued with this compound in combination with GSK2141795. mycancergenome.org
In uveal melanoma, a randomized trial compared this compound alone versus this compound combined with the Akt inhibitor GSK2141795, revealing no difference in median progression-free survival and only one partial response in each group. researchgate.net
Preclinical studies in meningioma have shown that MEK inhibition leads to PI3K/AKT activation, and co-targeting both pathways with a combination of the PI3K inhibitor alpelisib and this compound provided an additive effect on inhibiting cell viability. mdpi.com
A phase II clinical trial is investigating the clinical effect of this compound combined with everolimus and lenvatinib in the treatment of recurrent/refractory advanced solid tumors. careboxhealth.comclinicaltrial.beveeva.com
Combination with Pazopanib in Thyroid Cancer
Preclinical findings support the evaluation of this compound, alone or in combination with pazopanib or other kinase inhibitors, in thyroid cancer clinical trials. spandidos-publications.comresearchgate.net Studies have shown that the combination of this compound and pazopanib resulted in synergistic inhibition of tumor growth in thyroid cancer cell lines and xenograft models. spandidos-publications.comresearchgate.net
A phase I study evaluated the safety and efficacy of pazopanib and this compound in advanced solid tumors with an expansion cohort in differentiated thyroid cancer (DTC). nih.govgisttrials.org The combination showed clinical activity in DTC but did not achieve the prespecified response rate target. nih.gov In this study, objective response in the DTC cohort was 33%, and median progression-free survival was 10.7 months. nih.gov NRAS mutation was associated with response in this cohort. nih.gov
Table 1: Pazopanib and this compound Combination in Differentiated Thyroid Cancer (Phase I Expansion Cohort)
| Endpoint | Result |
| Objective Response Rate (ORR) | 33% |
| Median Progression-Free Survival (PFS) | 10.7 months |
Combination with Naporafenib in NRAS-Mutant Melanoma
The combination of naporafenib and this compound is being investigated for the treatment of unresectable or metastatic NRAS-mutant melanoma. A global, randomized, open-label Phase III study, SEACRAFT-2, is assessing the efficacy and safety of naporafenib administered with this compound compared to physician's choice of therapy (dacarbazine, temozolomide, or this compound monotherapy) in patients who have progressed on or are intolerant to a PD-1/PD-L1–based regimen. clinicaltrial.bemskcc.org
Combination with GNS561 in KRAS-Mutated Cholangiocarcinoma
A Phase 1b/2a study is combining GNS561 with this compound for advanced cholangiocarcinoma with a KRAS mutation. cancer.govclinicaltrials.eufroedtert.com This study aims to assess the safety and efficacy of this combination. froedtert.com Data from the ongoing Phase 1b portion is expected by year-end 2025 to support dose selection for Phase 2.
Combination with Azacitidine in Juvenile Myelomonocytic Leukemia
The combination of this compound and azacitidine is being investigated for the treatment of juvenile myelomonocytic leukemia (JMML). Preclinical studies have shown potential synergy between these two agents. ashpublications.org Researchers have found that the hypomethylating agent azacitidine plus the MEK inhibitor this compound may be a promising new combination to treat patients with JMML, according to a preclinical study. ascopost.com This combination may be capable of reducing some of the cancerous features of the disease by decreasing the number of cancerous blood stem cells and reducing the activity of the RAS/MAPK pathway. ascopost.com
A risk-stratified, non-randomized phase I/II clinical trial (T2020-004) is designed to examine the safety and efficacy of this compound in combination with other agents for children and young adults with newly diagnosed JMML. ashpublications.orgusc.educancer.gov In this trial, patients with lower-risk JMML will be treated with azacitidine in combination with this compound. ashpublications.orgusc.educancer.gov Patients with high-risk JMML will receive azacitidine and this compound in combination with chemotherapy. usc.educancer.gov This trial represents the first risk-stratified treatment strategy for patients with JMML and the first to use this combination of azacitidine and this compound. ashpublications.org
Combination with Anti-BRD4 Drugs for Resistance Prevention
Research has explored combining this compound with anti-BRD4 drugs to overcome or prevent resistance to MEK inhibition. In triple-negative breast cancer cells treated with this compound, resistance involved the formation of new enhancers bound by molecules like BRD4, which help turn on genes. unclineberger.orgunc.edu Pairing an investigational anti-BRD4 drug with this compound helped to stop tumor growth in experiments in cells and mouse models. unclineberger.orgunc.edu This combination was able to inhibit the reprogramming of tumor cells, potentially preventing or reversing the development of resistance to this compound. unclineberger.orgunc.edu
Studies in NF1-deficient ovarian cancer cell lines showed that MEK inhibitors were not durably effective due to kinome reprogramming. nih.gov MEK inhibitor-mediated destabilization of FOSL1 led to induced expression of receptor tyrosine kinases (RTKs) and their downstream signaling, contributing to overcoming MEK inhibitor therapy. nih.gov Inhibition of BET proteins using BET bromodomain inhibitors (BETi) blocked MEK inhibitor-induced RTK reprogramming, indicating that BRD2 and BRD4 are potential therapeutic targets in combination with MEK inhibitors to block resistance due to kinome reprogramming. nih.gov
Evaluation in Specific Cancer Types
This compound has been evaluated in various cancer types, primarily those with alterations in the RAS/RAF/MEK pathway. It is approved as a single agent or in combination with dabrafenib for the treatment of unresectable or metastatic melanoma with BRAF V600E or V600K mutations. drugbank.comcuremelanoma.org The combination with dabrafenib is also approved for metastatic non-small cell lung cancer and anaplastic thyroid cancer with BRAF V600E mutation. drugbank.com
Clinical trials are ongoing to investigate this compound in other solid tumors and in combination with other agents. cancer.govfda.gov This includes studies in pediatric patients with relapsed/refractory solid tumors, including neuroblastoma, NF1-associated plexiform neurofibromas, and gliomas. fda.gov
A phase II trial investigated this compound in patients with advanced BRAF V600 /NRAS Q61 wild-type, ICI-refractory melanoma. nih.govascopubs.org The combination of this compound plus low-dose dabrafenib demonstrated encouraging efficacy in these patients. nih.govascopubs.org
A phase I/II study is evaluating the combination of the AKT inhibitor GSK2141795 with dabrafenib and this compound in patients with BRAF mutant cancer. swog.org This study was terminated. swog.org
A trial is investigating GSK2256098 (a FAK inhibitor) alongside this compound for solid tumors that are not responding to other treatments, including an evaluation in mesothelioma of the lung. cancerresearchuk.org
Metastatic Melanoma (BRAF V600E/K-Mutated, NRAS-Mutant, BRAF/NRAS Wild-Type)
This compound has played a significant role in the treatment of metastatic melanoma, both as a monotherapy and, notably, in combination with the BRAF inhibitor dabrafenib. Aberrant MAPK pathway signaling is a hallmark of melanoma. nih.gov
In patients with BRAF V600E or BRAF V600K mutated unresectable or metastatic melanoma, the combination of dabrafenib and this compound has shown significant superiority in terms of objective response rate (ORR) and progression-free survival (PFS) compared to dabrafenib monotherapy. frontiersin.org A phase III study (METRIC) evaluating this compound monotherapy in patients with metastatic BRAF V600E or BRAF V600K mutations demonstrated improved response rates and PFS compared to chemotherapy. This compound achieved a 22% response rate and a median PFS of 4.8 months, versus 8% and 1.5 months for chemotherapy, respectively. nih.govmdpi.com Updated analysis of the METRIC trial showed a median PFS of 4.9 months and a median OS of 15.6 months for this compound monotherapy, compared to 1.5 months PFS and 11.3 months OS in the chemotherapy group. mdpi.com
For NRAS-mutant melanoma, MEK inhibition with this compound may have a role. In a phase I clinical trial, this compound was associated with stable disease in 2 out of 7 treated patients. nih.gov A phase I study combining this compound with paclitaxel in BRAF wild-type melanoma, which included patients with NRAS-mutant melanoma, demonstrated a 40% partial response rate, with 4 out of 6 patients with NRAS-mutant melanoma experiencing a response. nih.gov
In BRAF wild-type advanced melanoma that has progressed after immunotherapy, a retrospective study of this compound treatment in 22 patients (20 with NRAS mutations, 1 with NF1, 1 with GNA11) showed disease control in 36% of patients (6 stable disease, 2 partial responses). The median PFS was 2 months, and median OS was 6.5 months. researchgate.net Preclinical evidence suggests activity of this compound in BRAF/NRAS wild-type melanoma cell lines, including those with loss of NF1. nih.gov An early phase I study of this compound monotherapy resulted in partial responses (10%) in wild-type melanoma patients. nih.gov A phase II trial investigating this compound with or without low-dose dabrafenib in advanced, previously treated BRAF V600/NRAS Q61 wild-type melanoma showed a confirmed ORR of 29.2% with the combination. The median PFS was 13.3 weeks, and median OS was 54.3 weeks. nih.govresearchgate.net
Table 1: Summary of this compound Efficacy in Metastatic Melanoma
| Melanoma Mutation Status | Treatment Regimen | Study Type/Phase | ORR (%) | Median PFS | Median OS | Citation Indices |
| BRAF V600E/K-Mutated | This compound Monotherapy | Phase III (METRIC) | 22 | 4.8 months | 15.6 months | nih.govmdpi.com |
| BRAF V600E/K-Mutated | Dabrafenib + this compound | Phase I/II | 76 | 9.4 months | Not specified | onclive.com |
| NRAS-Mutant | This compound Monotherapy | Phase I | Not specified (Stable Disease) | Not specified | Not specified | nih.gov |
| NRAS-Mutant | This compound + Paclitaxel | Phase I | 40 | Not specified | 14 months | nih.gov |
| BRAF/NRAS Wild-Type (ICI-refractory) | This compound + Low-dose Dabrafenib | Phase II | 29.2 | 13.3 weeks | 54.3 weeks | nih.govresearchgate.net |
| BRAF V600 Wild-Type (Retrospective) | This compound Monotherapy (primarily NRAS) | Retrospective | 9 (PR) | 2 months | 6.5 months | researchgate.net |
Non-Small Cell Lung Cancer (BRAF V600E-Mutated)
The combination of dabrafenib and this compound has demonstrated efficacy in the treatment of BRAF V600E-mutated non-small cell lung cancer (NSCLC). theoncologynurse.comnih.gov A multicenter, open-label phase 2 study (BRF113928) evaluated the combination in both pretreated and treatment-naïve patients with metastatic BRAF V600E-mutant NSCLC. theoncologynurse.com
A single-arm, open-label, multicentre phase II study (NCT04452877) in Chinese patients with BRAF V600E mutation-positive metastatic NSCLC treated with dabrafenib plus this compound reported an ORR of 75%. amegroups.org At the data cut-off, the median duration of response, PFS, and OS were not reached. amegroups.org At 4 months, 90.0% of patients showed a response, and 91.7% achieved PFS. amegroups.org
Table 2: Summary of Dabrafenib + this compound Efficacy in BRAF V600E-Mutated NSCLC
| Patient Population | Study Type/Phase | ORR (%) | Median PFS | Median OS | 3-Year OS (%) | Citation Indices |
| Treatment-Naïve | Phase II | Not specified | Not specified | 17.3 months | 40 | theoncologynurse.com |
| Pretreated | Phase II | Not specified | Not specified | 18.2 months | 33 | theoncologynurse.com |
| Overall (Phase II) | Phase II | 63.2 | Not specified | Not specified | Not specified | dovepress.com |
| Chinese Patients | Phase II | 75 | Not reached | Not reached | Not specified | amegroups.org |
Thyroid Cancer (Anaplastic, BRAF-Mutated)
The combination of dabrafenib plus this compound has received approval in several countries for the treatment of BRAF V600E-mutant anaplastic thyroid cancer (ATC). uax.com This was based on data from the phase II Rare Oncology Agnostic Research (ROAR) basket study. uax.comnih.gov
An updated analysis of the ROAR ATC cohort, including 36 patients with unresectable or metastatic BRAF V600E-mutant ATC, demonstrated meaningful clinical activity. uax.comnih.gov The investigator-assessed ORR was 56%, including three complete responses. uax.comnih.gov The 12-month duration of response rate was 50%. uax.comnih.gov Median PFS was 6.7 months, and median OS was 14.5 months. uax.comnih.gov The 12-month PFS and OS rates were 43.2% and 51.7%, respectively, and the 24-month OS rate was 31.5%. uax.comnih.gov These survival rates are notable given the historically poor median OS of less than 6 months in patients with ATC. uax.comnih.gov
A retrospective study evaluating dabrafenib and this compound in 27 patients with metastatic BRAF V600E-mutated papillary thyroid cancer (PTC), including eight with poorly differentiated or anaplastic transformation, reported an ORR of 73.1% and a disease control rate (DCR) of 92.3%. e-crt.org Median PFS and OS were both 21.7 months in this cohort. e-crt.org
Table 3: Summary of Dabrafenib + this compound Efficacy in BRAF V600E-Mutated Anaplastic Thyroid Cancer
| Study | Patient Population | ORR (%) | Median PFS | Median OS | 12-Month OS Rate (%) | Citation Indices |
| ROAR Basket Study | Unresectable/Metastatic BRAF V600E ATC | 56 | 6.7 months | 14.5 months | 51.7 | uax.comnih.gov |
Colorectal Cancer (BRAF-Mutant, KRAS-Mutated)
The RAS/RAF/MEK/ERK signaling pathway is critical in colorectal cancers, and KRAS, NRAS, and BRAF mutations are common. aacrjournals.org Colorectal cancers harboring KRAS, NRAS, or BRAF-activating mutations are known to have worse prognoses. aacrjournals.org
Single-agent MEK inhibition has shown limited activity in BRAF-mutant metastatic colorectal cancer (mCRC). An early phase I study of this compound in patients with treatment-refractory KRAS- or BRAF-mutant advanced CRC did not identify an objective response in 28 patients. amegroups.org
Combination strategies targeting the MAPK pathway have been explored in BRAF-mutant CRC. A phase I/II trial of dabrafenib and this compound in 36 patients with BRAF-mutated CRC found that 1 patient achieved a complete response, and 3 had an unconfirmed partial response. onclive.com Another study of dabrafenib and this compound in 43 patients with BRAF-mutant CRCs reported disease control in 27 patients (22 stable disease, 4 partial response, 1 complete response). dovepress.com
Preclinical studies have investigated combining this compound with other agents in KRAS-mutated CRC. High-throughput screening identified paclitaxel as synergistic with this compound in KRAS-mutated CRC spheroids and cell lines, enhancing cell death and inhibiting tumor growth in patient-derived xenografts. nih.gov These preclinical findings support further clinical investigation of this combination in KRAS-mutated mCRC. nih.gov
Multiple Myeloma
Activating mutations of the MAPK pathway are reported in over half of myeloma tumors. researchgate.net While experience with MEK inhibitors in solid tumors suggests that tumors harboring BRAF or RAS mutations are more likely to respond, response rates and duration of responses can be limited. researchgate.net
Early anecdotal activity with the combination of dabrafenib and this compound has been noted in multiple myeloma. frontiersin.org A retrospective analysis identified 58 this compound-treated relapsed/refractory multiple myeloma (RRMM) cases, of which 51 harbored KRAS, NRAS, or BRAF mutations. frontiersin.org Single-agent this compound showed early success in isolated individuals, including one KRAS-mutated patient with multiply relapsed myeloma who achieved an impressive response. frontiersin.org
Oral Cavity Squamous Cell Carcinoma (OCSCC)
Ras/MEK/ERK pathway activation is common in oral cavity squamous cell carcinoma (OCSCC). aacrjournals.orgnih.govaacrjournals.org A phase II neoadjuvant window-of-opportunity clinical trial evaluated this compound in patients with stage II–IV OCSCC prior to surgery. aacrjournals.orgnih.govaacrjournals.orgresearchgate.net
In this study, this compound administration led to a significant reduction in Ras/MEK/ERK pathway activation and a decrease in CD44 expression. aacrjournals.orgnih.gov A clinical tumor response, based on modified World Health Organization criteria, was observed in 11 out of 17 (65%) evaluable patients, with a median decrease of 46%. aacrjournals.orgnih.govresearchgate.net A partial metabolic response (≥25% reduction in SUVmax) was observed in 6 out of 13 (46%) evaluable patients, with a median decrease of 25%. aacrjournals.orgnih.govresearchgate.net Clinical-to-pathologic tumor downstaging occurred in 9 out of 17 (53%) evaluable patients. aacrjournals.orgnih.govresearchgate.net These findings suggest that this compound is a promising therapeutic option for OCSCC patients. nih.govbohrium.com
Table 4: Summary of this compound Efficacy in Neoadjuvant OCSCC
| Endpoint | Response/Decrease | Number of Evaluable Patients | Percentage (%) | Citation Indices |
| Clinical Tumor Response (by WHO criteria) | Observed | 17 | 65 | aacrjournals.orgnih.govresearchgate.net |
| Clinical Tumor Measurement Decrease (median) | 46% | Not specified | Not specified | aacrjournals.orgnih.govresearchgate.net |
| Partial Metabolic Response (≥25% SUVmax reduction) | Observed | 13 | 46 | aacrjournals.orgnih.govresearchgate.net |
| SUVmax Decrease (median) | 25% | Not specified | Not specified | aacrjournals.orgnih.govresearchgate.net |
| Clinical-to-Pathologic Tumor Downstaging | Occurred | 17 | 53 | aacrjournals.orgnih.govresearchgate.net |
High-Grade Gliomas (Pediatric and Young Adult)
Gliomas are the most common primary brain and CNS tumors in children and adolescents, with pediatric high-grade gliomas (pHGGs) accounting for a significant proportion. ascopubs.org The BRAF V600 mutation is detected in 5%-10% of pHGGs. ascopubs.orgesmo.org
The combination of dabrafenib plus this compound has shown promising results in pediatric and young adult patients with relapsed/refractory BRAF V600-mutated high-grade gliomas. ascopubs.orgesmo.orgnih.gov A phase II study evaluated this combination in 41 pediatric patients with previously treated BRAF V600-mutant HGG. ascopubs.orgesmo.org
The independently assessed ORR was 56%. ascopubs.orgesmo.org The median duration of response (DoR) was 22.2 months. ascopubs.orgesmo.org Median OS was 32.8 months. ascopubs.org These results compare favorably with historical data from molecularly unselected cohorts of relapsed/refractory pHGG. ascopubs.orgesmo.org Younger age, BRAF V600 mutation, and longer therapy periods were associated with better radiological outcomes in pediatric patients. nih.gov
A systematic review and meta-analysis comparing outcomes of dabrafenib plus this compound in pediatric and adult gliomas (including low-grade gliomas) with BRAF V600 mutations found a higher, though not statistically significant, ORR (53% vs. 39%) and clinical benefit rate (87% vs. 73%) in pediatric patients compared to adults. nih.gov
Table 5: Summary of Dabrafenib + this compound Efficacy in Relapsed/Refractory BRAF V600-Mutated Pediatric High-Grade Glioma
| Study Type/Phase | Patient Population | ORR (%) | Median DoR | Median OS | Citation Indices |
| Phase II | Relapsed/Refractory Pediatric BRAF V600-Mutated HGG | 56 | 22.2 months | 32.8 months | ascopubs.orgesmo.org |
Cholangiocarcinoma
Cholangiocarcinoma (CCA), a cancer of the bile ducts, is often diagnosed at advanced stages with limited treatment options. mdanderson.orgbjmo.be Approximately 5% of biliary tract tumors harbor BRAF V600E mutations, which activate the MAPK pathway. bjmo.bemdpi.com The combination of dabrafenib (a BRAF inhibitor) and this compound has shown promise in patients with BRAF V600E-mutated CCA. mdanderson.orgbjmo.betargetedonc.com
Another phase IIa study investigated this compound alone in 20 Japanese patients with advanced biliary tract cancers refractory to gemcitabine-based therapy. withpower.comnih.gov The 12-week non-progression rate was 10%, which did not meet the target threshold. withpower.comnih.gov The median PFS was 10.6 weeks. nih.gov
Ongoing research continues to explore this compound in CCA, including in combination with other agents. A Phase 1b/2a study (NCT04296945) is evaluating GNS561 in combination with this compound in advanced KRAS-mutated CCA. uvahealth.com Preclinical studies also suggest that this compound may improve the immunogenicity of tumor cells, potentially enhancing the efficacy of combination therapy with anti-PD-1 antibodies in intrahepatic CCA models. nih.gov
| Study (NCT Identifier) | Patient Population (BRAF V600E-mutated CCA) | Intervention | ORR (Investigator-Assessed) | Median DoR (months) | Median PFS (months) | Median OS (months) |
| ROAR (NCT02034110) | 43 patients (previously treated) | Dabrafenib + this compound | 51% (95% CI 36–67) | 8.7 | 9.1 | 13.5 |
Juvenile Myelomonocytic Leukemia
Juvenile myelomonocytic leukemia (JMML) is a rare and aggressive childhood myeloproliferative disorder characterized by aberrant signaling through the Ras pathway, often due to mutations in genes such as PTPN11, KRAS, NRAS, and NF1. ashpublications.orgaacrjournals.orgaacr.org Hematopoietic stem cell transplantation (HSCT) is a potentially curative treatment, but outcomes are poor for patients with relapsed or refractory disease. ashpublications.orgaacrjournals.org
This compound, as a MEK inhibitor targeting the Ras/MAPK pathway, has been investigated in pediatric patients with JMML. ashpublications.orgaacrjournals.orgaacr.org A phase II clinical trial (ADVL1521, NCT03190915) conducted by the Children's Oncology Group evaluated this compound monotherapy in children with relapsed or refractory JMML. ashpublications.orgaacrjournals.orgaacr.orgnih.gov Ten patients were enrolled, and the objective response rate was 50%, including two complete responses and three partial responses. aacrjournals.orgaacr.orgnih.gov Seven of the 10 patients were alive after a median follow-up of two years. aacrjournals.orgaacr.orgnih.gov Four patients with refractory disease proceeded to HSCT after receiving this compound. aacrjournals.orgnih.gov Three patients completed the maximum 12 cycles and continued on off-protocol this compound without HSCT. aacrjournals.orgnih.gov
A phase 1/2 non-randomized study (T2020-004, NCT05849662) is evaluating this compound in combination with azacitidine in newly diagnosed lower-risk JMML patients and in combination with azacitidine, fludarabine, and cytarabine in high-risk patients. ashpublications.orgcincinnatichildrens.orgcancer.gov Preclinical studies using JMML patient-derived xenograft mouse models have also demonstrated that this compound monotherapy significantly prolongs survival by inhibiting the RAS pathway. tandfonline.comnih.gov
| Study (NCT Identifier) | Patient Population (JMML) | Intervention | Objective Response Rate | Overall Survival (at median 2 years) |
| ADVL1521 (NCT03190915) | 10 patients (relapsed/refractory) | This compound monotherapy | 50% | 70% |
Triple-Negative Breast Cancer
Triple-negative breast cancer (TNBC) is an aggressive subtype of breast cancer that lacks expression of estrogen receptor, progesterone receptor, and HER2, limiting targeted therapy options. researchgate.netega-archive.orgaacrjournals.org Preclinical data suggest that activation of the RAS/MEK/ERK pathway may contribute to chemotherapy resistance in TNBC. aacrjournals.org
Research has explored the potential of targeting the MEK pathway with this compound in TNBC. A phase II study (OSU 13317) investigated this compound alone and in combination with the AKT inhibitor uprosertib in patients with metastatic TNBC previously treated with chemotherapy. aacrjournals.orgoncodaily.com In the this compound alone arm, 2 out of 37 enrolled patients had a partial response, and 6 had stable disease. aacrjournals.org In the combination arm (19 evaluable patients), 3 patients had a partial response and 3 had stable disease. aacrjournals.org While this compound monotherapy showed limited efficacy, the combination with uprosertib was associated with a numerically greater objective response rate. oncodaily.com
Studies have also investigated mechanisms of resistance to this compound in TNBC. Researchers have found that TNBC cells can develop resistance by altering their genetic landscape and forming new enhancers that turn on alternative protein pathways. unclineberger.org Combining this compound with an anti-BRD4 drug in laboratory models helped to inhibit this reprogramming and prevent the onset of resistance. unclineberger.org Preclinical studies have also shown that ONC201 and this compound synergistically inhibit the growth of TNBC cells. researchgate.net
Emerging Research Areas in Non-Oncologic Applications
Treatment of Noonan Syndrome-Associated Cardiovascular Manifestations
Noonan syndrome (NS) is a genetic disorder caused by mutations in genes involved in the Ras/MAPK signaling pathway, including PTPN11, KRAS, SOS1, and RAF1. frontiersin.orgradboudumc.nlnih.gov These mutations lead to an overactivation of the pathway and can result in various clinical features, including severe cardiovascular anomalies such as hypertrophic cardiomyopathy (HCM) and pulmonary valve stenosis (PVS). frontiersin.orgradboudumc.nlnih.govmdpi.com
Given the role of the hyperactive Ras/MAPK pathway in the pathogenesis of NS-associated cardiovascular manifestations, MEK inhibitors like this compound have been explored as a potential targeted therapy. frontiersin.orgradboudumc.nlnih.govjacc.org Retrospective studies and case reports have described the use of this compound in infants and children with severe NS-associated cardiac and lymphatic issues. frontiersin.orgradboudumc.nlnih.govchusj.orgnih.gov
A retrospective study analyzing the clinical records of 61 NS patients with thickening of the heart muscle showed that children who received compassionate use of this compound in addition to standard treatment had a lower risk of death, heart transplantation, or other major heart surgeries compared to those receiving standard treatment alone. radboudumc.nlchusj.org Over a three-year period, the probability of these events was 20% for children treated with this compound, compared to 80% for those on standard treatment. chusj.org The study also indicated that the cardiac status of patients treated with this compound improved. radboudumc.nl
Case reports have also documented positive responses to this compound in severe cases. For instance, a preterm infant with congenital pulmonary lymphangiectasis and chylothorax refractory to conventional management showed resolution of chylothorax and gradual pulmonary function improvement after a five-week course of this compound. frontiersin.orgnih.govnih.gov this compound treatment has also been associated with regression of left ventricular hypertrophy and improved cardiac function in patients with NS-related HCM. jacc.orgahajournals.org
While formal clinical trials of this compound specifically for neonatal/pediatric NS-associated cardiovascular manifestations are limited, the available evidence from retrospective studies and case series suggests that this compound is a promising drug in this setting. frontiersin.orgnih.govnih.gov Further clinical trials are considered warranted to establish standardized protocols and confirm efficacy and safety. frontiersin.orgnih.govnih.govresearchgate.net Preclinical studies in mouse models of NS have also demonstrated that this compound can improve the cardiovascular phenotype, including a decrease in heart weight-to-body weight ratio and reduced proliferating cells in the heart. thrasherresearch.org
| Condition (Noonan Syndrome-Associated) | Manifestation(s) Studied | Research Type | Key Findings |
| Noonan Syndrome | Hypertrophic Cardiomyopathy, Lymphatic Anomalies (Chylothorax) | Retrospective studies, Case reports, Preclinical models | Improved cardiac status, reduced risk of major cardiac events, resolution of chylothorax, regression of LVH, improved cardiovascular phenotype in mouse models. frontiersin.orgradboudumc.nlnih.govchusj.orgnih.govahajournals.orgthrasherresearch.org |
Pharmacology and Pharmacokinetics Research
Absorption and Bioavailability Characteristics
Following oral administration, trametinib is rapidly absorbed. drugbank.comdovepress.com The median time to achieve peak plasma concentrations (Tmax) is approximately 1.5 hours under fasted conditions. drugbank.comdovepress.comtga.gov.au The mean absolute bioavailability of a single 2 mg oral tablet dose is reported to be around 72% relative to an intravenous microdose. drugbank.comdovepress.comtga.gov.auresearchgate.net
Studies have shown that both the maximum plasma concentration (Cmax) and the area under the concentration-time curve (AUC) increase in a dose-proportional manner following repeat daily dosing of this compound tablets ranging from 0.125 mg to 4 mg. drugbank.com this compound accumulates with repeat daily dosing, with a mean accumulation ratio of approximately 6.0 at a 2 mg once daily dose, reaching steady-state by approximately Day 15. drugbank.comhres.ca
Food intake can impact this compound absorption. Administration of a single 2 mg dose with a high-fat, high-calorie meal resulted in a significant decrease in Cmax (approximately 70%) and a reduction in AUC (ranging from 10% to 24%) compared to administration under fasted conditions. drugbank.comtga.gov.aufda.govnovartis.com The Tmax was also delayed by approximately four hours when taken with a high-fat meal. drugbank.com
This compound is highly bound to human plasma proteins, with approximately 97.4% protein binding. drugbank.comhres.ca The apparent volume of distribution (Vc/F) is estimated to be 214 L. drugbank.com
Table 1: this compound Absorption and Bioavailability Parameters (Fasted Conditions)
| Parameter | Value | Source |
| Tmax (median) | 1.5 hours | drugbank.comdovepress.comtga.gov.au |
| Absolute Bioavailability (2 mg tablet) | ~72% | drugbank.comdovepress.comtga.gov.auresearchgate.net |
| Protein Binding | ~97.4% | drugbank.comhres.ca |
| Apparent Volume of Distribution (Vc/F) | 214 L | drugbank.com |
| Accumulation Ratio (2 mg QD) | ~6.0 | drugbank.comhres.ca |
| Steady-State Achieved | By Day 15 | drugbank.comhres.ca |
Metabolism and Elimination Pathways
This compound is metabolized predominantly via deacetylation. drugbank.combccancer.bc.canih.govhres.ca This process is primarily mediated by carboxylesterases (specifically carboxylesterase 1b/c and 2) and other hydrolytic enzymes. drugbank.comhres.ca In addition to deacetylation, in vitro findings suggest that metabolism may also involve mono-oxygenation, hydroxylation, and glucuronidation, either alone or in combination with deacetylation. drugbank.comnih.govhres.canih.gov CYP3A4-mediated oxidation is considered a minor metabolic pathway. drugbank.com
The primary route of elimination for this compound and its metabolites is through feces. drugbank.comnih.govhres.canih.gov Following oral administration of [14C]-trametinib, over 80% of the excreted radioactivity was recovered in the feces. drugbank.comnih.govhres.ca Urinary excretion accounts for a minor portion, less than 20% of the excreted radioactivity, with less than 0.1% of the excreted dose recovered as the parent molecule in urine. drugbank.combccancer.bc.canih.govhres.ca
The estimated elimination half-life of this compound ranges from 3.9 to 5.3 days. drugbank.combccancer.bc.cadovepress.comnih.govhres.ca The apparent clearance is approximately 4.9 L/h. drugbank.comnih.gov
Table 2: this compound Metabolism and Elimination Summary
| Pathway | Description | Primary Enzymes/Mediators |
| Metabolism | Predominantly Deacetylation, also Mono-oxygenation, Hydroxylation, Glucuronidation | Carboxylesterases (1b/c, 2), Other Hydrolytic Enzymes, (Minor CYP3A4) |
| Primary Metabolite | Deacetylated metabolite (M5) | |
| Major Excretion Route | Feces (>80% of excreted radioactivity) | |
| Minor Excretion Route | Urine (<20% of excreted radioactivity) | |
| Elimination Half-life | 3.9 - 5.3 days | |
| Apparent Clearance | ~4.9 L/h |
Impact of Hepatic and Renal Impairment on Systemic Exposure
The impact of hepatic and renal impairment on the pharmacokinetics of this compound has been evaluated. Based on population pharmacokinetic analyses and clinical data, mild hepatic impairment does not appear to significantly affect this compound oral clearance or systemic exposure compared to patients with normal hepatic function. hres.cafda.govnovartis.comeuropa.eunih.gov However, there is limited clinical data available for patients with moderate or severe hepatic impairment, and caution is advised in these populations. hres.caeuropa.euresearchgate.net
Similarly, renal impairment is considered unlikely to have a clinically relevant effect on this compound pharmacokinetics due to the low renal excretion of the drug. hres.canovartis.comeuropa.eu Population pharmacokinetic analyses characterizing this compound in patients with mild and moderate renal impairment have shown no significant effect on oral clearance or systemic exposure. hres.canovartis.comeuropa.eu Data for patients with severe renal impairment are not available. hres.canovartis.comeuropa.eu
Population Pharmacokinetic Analyses and Variability in Exposure
Population pharmacokinetic analyses have been conducted to characterize this compound pharmacokinetics and identify potential factors contributing to variability in exposure. These analyses have shown that this compound plasma concentrations can be described using a two-compartment model. nih.govnih.gov
Factors identified as influencing this compound oral clearance include gender and body weight. hres.canovartis.comnih.gov Female patients, particularly those with lower body weights, have been observed to have lower this compound clearance and consequently higher systemic exposure compared to male patients. hres.canovartis.comnih.gov For instance, at a median weight of 79 kg, female patients had approximately 21% lower clearance and 25% higher AUC than males. hres.canovartis.com
Age has generally been found to have no relevant clinical effect on this compound pharmacokinetics based on population analyses. hres.canovartis.comnih.gov However, one study identified a negative effect of age on clearance, explaining a portion of the associated variability. nih.gov This study also suggested that concentrations might be higher in patients over 65 years of age. nih.gov
Drug-Drug Interaction Potential
Based on in vitro and limited in vivo data, this compound is generally considered unlikely to significantly affect the pharmacokinetics of other medicinal products through interactions with CYP enzymes or transporters. europa.eumedicalletter.orgcancer-druginteractions.org this compound is not a substrate of major CYP enzymes (such as CYP3A4) or common transporters like BCRP, OATP1B1, OATP1B3, OATP2B1, OCT1, MRP2, and MATE1. fda.goveuropa.eu
While this compound is an in vitro substrate of BSEP and the efflux transporter P-gp, significant changes in this compound exposure due to inhibition of BSEP are considered unlikely. fda.goveuropa.eu
Drug-drug interactions mediated by the hydrolytic enzymes responsible for this compound deacetylation cannot be definitively ruled out and could potentially influence this compound exposure. europa.eu
Although this compound itself is not a significant inhibitor or inducer of major CYP enzymes, caution is advised when co-administering this compound in combination with other drugs that have a known risk of QTc prolongation, particularly when used in combination with dabrafenib, as the combination has shown higher incidences of QTc increases compared to monotherapy. cancer-druginteractions.org
Table 3: Factors Influencing this compound Pharmacokinetics (Population PK)
| Factor | Effect on Clearance | Effect on Exposure (AUC) | Notes | Source |
| Gender (Female vs Male) | Lower | Higher | More pronounced in lower body weight females | hres.canovartis.comnih.gov |
| Body Weight | Increases Clearance | Decreases Exposure | Positive correlation with clearance | nih.gov |
| Age | No significant effect (generally) | No significant effect (generally) | Some analysis suggests negative effect on clearance in older patients | hres.canovartis.comnih.govnih.gov |
| Renal Impairment (Mild/Moderate) | No significant effect | No significant effect | Low renal excretion of parent drug | hres.canovartis.comeuropa.eu |
| Hepatic Impairment (Mild) | No significant effect | No significant effect | Limited data for moderate/severe | hres.cafda.govnovartis.comeuropa.eunih.gov |
Table 4: this compound Drug Interaction Potential Summary
| Interaction Mechanism | This compound Effect | Potential Impact on Co-administered Drugs | Source |
| CYP Enzyme Inhibition/Induction | Unlikely to significantly inhibit or induce major CYPs | Low potential for impact | europa.eumedicalletter.orgcancer-druginteractions.org |
| Transporter Interactions | In vitro substrate of BSEP, P-gp; Not substrate of others | Unlikely to significantly affect exposure of BSEP substrates; Potential for transient inhibition of BCRP substrates in the gut | fda.goveuropa.eucancer-druginteractions.org |
| Hydrolytic Enzyme Interactions | Metabolized by hydrolytic enzymes | Potential for influence on this compound exposure | europa.eu |
| QTc Prolongation Risk | Low risk as monotherapy; Increased risk in combination with dabrafenib | Caution with co-administration of drugs with known QTc risk | cancer-druginteractions.org |
Biomarkers in Trametinib Research
Predictive Biomarkers for Response
Predictive biomarkers help identify patients most likely to benefit from Trametinib treatment. These often involve alterations within the MAPK pathway itself or markers reflecting the downstream effects of MEK inhibition.
BRAF V600E/K Mutations
Mutations in the BRAF gene, particularly the V600E and V600K alterations, are well-established predictive biomarkers for response to this compound, especially in melanoma. nih.govmycancergenome.orgnih.govmdpi.com These mutations lead to constitutive activation of BRAF, a kinase upstream of MEK in the MAPK pathway. aacrjournals.org this compound, by inhibiting MEK, blocks the downstream signaling driven by mutated BRAF. mdpi.com
NRAS Mutations
NRAS mutations are also being investigated as predictive biomarkers for this compound sensitivity. NRAS mutations are found in a significant subset of melanomas (15%-20%) and can also occur in other cancers like NSCLC (0.5-1%) and pediatric B-cell precursor acute lymphoblastic leukemia (BCP-ALL) (up to 44.2% of cases with RAS pathway mutations). nih.govsmw.chnih.govaacrjournals.org Preclinical data suggest that cell lines with NRAS mutations are sensitive to MEK inhibitors like this compound. smw.chaacrjournals.org
Clinical studies have explored the efficacy of MEK inhibitors in NRAS mutant melanoma. In a phase II study of binimetinib (another MEK inhibitor), a 20% partial response rate was observed in patients with NRAS mutant melanoma. nih.gov A phase I study combining this compound with paclitaxel in BRAF wild-type melanoma, which included NRAS mutant cases, showed a 40% partial response rate, with 4 out of 6 NRAS-mutant patients experiencing a response. nih.gov While NRAS mutations show promise as a predictive marker, further clinical validation is needed, particularly for this compound monotherapy. smw.ch
Dynamic Molecular Markers of Pathway Inhibition (e.g., p-ERK1/2, p27, cyclin D1)
Dynamic biomarkers, assessed after the initiation of treatment, can indicate whether this compound is effectively inhibiting the MAPK pathway. Phosphorylated ERK1/2 (p-ERK1/2) is a direct downstream target of MEK, and its reduction upon this compound treatment indicates effective pathway inhibition. nih.govaacrjournals.org Studies have shown that this compound induces rapid and sustained dephosphorylation of p-ERK1/2 in sensitive cell lines and tumor models. nih.gov In a clinical trial in oral cavity squamous cell carcinoma (OCSCC), this compound treatment resulted in a significant reduction in p-ERK1/2 expression in a subset of patients. aacrjournals.org
Changes in cell cycle regulatory proteins like p27 and cyclin D1 are also observed following MEK inhibition. This compound has been shown to induce p27 accumulation and downregulate cyclin D1, suggesting cell cycle arrest. nih.govnih.govnih.govresearchgate.net These changes reflect the impact of MAPK pathway inhibition on cell cycle progression. researchgate.net
Markers of Cellular Outcomes (e.g., Cleaved PARP, phospho-4E-BP1, phospho-AKT)
Beyond direct pathway inhibition, markers reflecting cellular outcomes like apoptosis or altered protein synthesis can predict response. Cleaved PARP is a marker of apoptosis. nih.govoup.comnih.govaacrjournals.org Studies in NSCLC cell lines showed that PARP cleavage occurred in a subset of lines sensitive to this compound, suggesting that apoptosis is a mechanism of response in some contexts. nih.gov
Phosphorylation of 4E-BP1 (phospho-4E-BP1) is involved in regulating protein synthesis, and its modulation can reflect the impact of MEK inhibition on this process. nih.govoup.comresearchgate.netresearchgate.net Increased phospho-4E-BP1 has been observed in some this compound-treated cells, suggesting varied cellular outcomes, including potentially senescence or autophagy. nih.govresearchgate.netresearchgate.net
Phosphorylated AKT (phospho-AKT) is a marker of the PI3K/AKT/mTOR pathway, which can be activated in response to MEK inhibition and contribute to resistance. oup.comresearchgate.netmdc-berlin.de While complex, the modulation of phospho-AKT levels following this compound treatment may provide insights into cellular responses and potential compensatory pathway activation. researchgate.net
MAPK Pathway-Activating Alterations
Beyond BRAF and NRAS mutations, other alterations that activate the MAPK pathway can potentially serve as predictive biomarkers for MEK inhibitor sensitivity. These include mutations in KRAS, NF1, and MAP2K1 (which encodes MEK1). smw.chnih.govaacrjournals.orgamegroups.orgmdpi.comascopubs.org Patients with activating mutations in canonical MAPK pathway components have been reported to respond more frequently to MEK inhibitors compared to those without such mutations. amegroups.org
Research in low-grade serous ovarian carcinoma (LGSOC) with KRAS, BRAF, or NRAS mutations showed markedly higher PFS and ORR with this compound compared to wild-type cases. amegroups.org Functional assays measuring the level of MAPK pathway activation induced by different RAS mutations have also shown correlation between higher activity scores and longer PFS after MEK inhibitor treatment. ascopubs.org
Biomarkers for Resistance Development
Despite initial responses, many patients treated with this compound develop acquired resistance. Identifying biomarkers associated with resistance is crucial for developing strategies to overcome it. Mechanisms of resistance often involve the reactivation of the MAPK pathway or the activation of alternative survival pathways, such as the PI3K/AKT/mTOR pathway. mdpi.commdc-berlin.denih.gov
Genetic alterations leading to resistance to BRAF inhibitors, which can also apply to MEK inhibitors like this compound, include NRAS mutation, BRAF amplification, MEK mutations, NF1 mutations, AKT amplification, and loss of PTEN. nih.gov Overexpression of receptor tyrosine kinases (RTKs) or other signaling molecules can also contribute to resistance by reactivating downstream pathways. nih.gov
In preclinical models, resistance to this compound has been associated with increased mTOR signaling, reflected by increased levels of phosphorylated AKT and 4E-BP1. oup.comnih.gov Loss of NF1 expression has also been linked to reduced sensitivity to MEK inhibition in glioblastoma cell lines. oup.com
Dynamic changes in biomarkers during treatment can also indicate the emergence of resistance. For example, the reappearance of p-ERK signaling or changes in the expression of cell cycle or survival markers despite continued this compound treatment can suggest resistance development.
Here is a summary table of biomarkers discussed:
| Biomarker Category | Examples | Role in this compound Research | Relevant Section |
| Predictive Biomarkers for Response | 7.1 | ||
| Genetic Mutations | BRAF V600E/K | Predicts sensitivity, especially in melanoma. | 7.1.1 |
| NRAS Mutations | Potential predictor, particularly in melanoma and other cancers. | 7.1.2 | |
| MAPK Pathway-Activating Alterations (e.g., KRAS, NF1, MAP2K1) | Indicates potential sensitivity to MEK inhibition. | 7.1.5 | |
| Dynamic Molecular Markers | p-ERK1/2 (decreased) | Indicates effective pathway inhibition. | 7.1.3 |
| p27 (increased) | Indicates cell cycle arrest. | 7.1.3 | |
| Cyclin D1 (downregulated) | Indicates cell cycle arrest. | 7.1.3 | |
| Markers of Cellular Outcomes | Cleaved PARP (increased) | Indicates apoptosis. | 7.1.4 |
| phospho-4E-BP1 (modulated) | Reflects altered protein synthesis, potential senescence/autophagy. | 7.1.4 | |
| phospho-AKT (modulated) | May indicate compensatory pathway activation. | 7.1.4 | |
| Biomarkers for Resistance Development | 7.2 | ||
| Genetic/Epigenetic Alterations | NRAS mutation, BRAF amplification, MEK mutations, NF1 mutations, AKT amplification, PTEN loss | Associated with acquired resistance. | 7.2 |
| Pathway Reactivation | Reactivation of MAPK or PI3K/AKT/mTOR pathways | Mechanisms of acquired resistance. | 7.2 |
Biomarkers for Toxicity Assessment
Research into biomarkers for this compound toxicity assessment focuses on identifying measurable indicators that correlate with the occurrence or severity of adverse events. These biomarkers can include circulating factors, genetic markers, or changes in protein expression levels.
Studies have investigated potential biomarkers for specific this compound-induced toxicities, such as cardiotoxicity and skin toxicity nih.govjacc.orgclinicaltrials.gov. Cardiotoxicity, including cardiomyopathy, has been observed in some patients treated with this compound nih.govjacc.org. Research in murine models of this compound-induced cardiotoxicity has identified differentially expressed genes and affected signaling pathways, including PI3K/AKT and JAK/STAT signaling nih.govjacc.org. Elevated markers of oxidative stress, myofibrillar degeneration, down-regulation of the apelin receptor, and connexin-43 mislocalization were also observed in cardiac tissue nih.govjacc.org. Transcriptomic analysis revealed 435 differentially expressed genes and 116 differential signaling pathways in cardiac tissue following this compound treatment in mice nih.govjacc.org. Upstream gene analysis suggested interleukin-6 as a potential regulator of some of these genes nih.govjacc.org.
Another area of investigation involves circulating biomarkers. Some studies have explored the potential of serum markers like soluble CD163 (sCD163) and chemokines such as CXCL5, CXCL9, CXCL10, and CXCL11 in the context of adverse events associated with targeted therapies, including MEK inhibitors researchgate.netresearchgate.net. While some findings relate to combination therapies or immune checkpoint inhibitors, they suggest a potential role for these inflammatory markers in predicting or indicating the onset of certain adverse events researchgate.netresearchgate.net. For instance, increased serum levels of sCD163 were observed in patients who developed adverse events in a study involving nivolumab, and there is a hypothesis that these soluble factors might correlate with adverse events in dabrafenib plus this compound combination therapy researchgate.netresearchgate.net. Increased levels of sCD163 were noted in patients experiencing pyrexia with dabrafenib and this compound, and elevated levels of CXCL9, CXCL10, and CXCL11 were prominent in patients with higher grade adverse events researchgate.net.
Biomarker studies are also being conducted to understand and predict skin toxicity associated with this compound, both as a monotherapy and in combination with dabrafenib clinicaltrials.gov. These studies aim to identify biomarkers in skin, blood, and tumor samples that correlate with the development of skin-related adverse events, such as rash and proliferative cutaneous lesions clinicaltrials.govascopubs.org.
Further research is needed to fully validate and establish the clinical utility of these potential biomarkers for predicting and managing this compound toxicity.
| Biomarker Category | Specific Biomarkers Investigated | Associated Toxicity | Research Findings (Examples) |
| Transcriptomic Changes | Differentially expressed genes, PI3K/AKT pathway, JAK/STAT pathway | Cardiotoxicity | 435 differentially expressed genes and 116 affected pathways identified in murine cardiac tissue; suggests involvement of PI3K/AKT and JAK/STAT signaling. nih.govjacc.org |
| Circulating Proteins | Soluble CD163 (sCD163), CXCL5, CXCL9, CXCL10, CXCL11 | Various (e.g., pyrexia, general AEs) | Increased sCD163 levels associated with adverse events; elevated CXCL9, CXCL10, CXCL11 with higher grade adverse events in combination therapy studies. researchgate.net |
| Tissue-Specific Markers | Oxidative stress markers, myofibrillar degeneration markers, apelin receptor, connexin-43 | Cardiotoxicity | Observed in murine cardiac tissue following this compound treatment. nih.govjacc.org |
| Cellular Changes | Immune cells (e.g., NK cells, dendritic cells, mast cells) | Cardiotoxicity | Changes in the proportion of certain immune cells observed in a dataset related to this compound-induced cardiotoxicity. biorxiv.org |
Mechanistic Insights into Adverse Events Associated with Trametinib
On-Target Toxicities Affecting Normal Cells
The core mechanism of trametinib involves inhibiting the activity of MEK1 and MEK2, thereby suppressing ERK signaling. jadpro.comnih.govcancercareontario.cadermnetnz.org This pathway, while hyperactive in certain cancers, is also fundamental for the growth, differentiation, inflammation, and survival of normal cells across various tissues. cancercareontario.cabccancer.bc.ca The inhibition of ERK signaling in these healthy cells constitutes "on-target" toxicity, directly resulting from the drug's intended mechanism of action. jadpro.comnih.gov These effects can manifest in different organ systems and often necessitate dose modifications to maintain a balance between therapeutic benefit and manageable side effects. jadpro.comnih.gov
Mechanisms of Pyrexia (Fever) in Combination Therapy
Pyrexia, or fever, is a frequently reported adverse event, notably more prevalent and sometimes more severe when this compound is administered in combination with dabrafenib compared to this compound monotherapy. jadpro.comnih.govnih.govresearchgate.neteuropa.eu The precise mechanism driving this combination-specific pyrexia remains under investigation. nih.govresearchgate.net However, current hypotheses suggest that this compound modulates the pyrexia process initiated by dabrafenib. nih.govresearchgate.net One proposed mechanism involves the paradoxical activation of the MAPK pathway in lymphocytes, drawing parallels to the febrile episodes seen in familial Mediterranean fever. researchgate.net Additionally, BRAF inhibition by dabrafenib is thought to stimulate inflammasome activation and the production of interleukin 1 beta (IL-1 beta) in dendritic cells, contributing to a pro-inflammatory state that can lead to fever. nih.gov
Cardiac Dysfunction (e.g., Left Ventricular Ejection Fraction Decrease, PR Prolongation)
Cardiac adverse events, including a decrease in left ventricular ejection fraction (LVEF) and PR interval prolongation, have been associated with this compound treatment. nih.govbccancer.bc.caeuropa.eudovepress.comcancercareontario.camedscape.comnih.govjacc.org The MAPK pathway is integral to normal cardiac function and possesses cardioprotective properties. nih.govjacc.org Inhibition of this pathway by this compound is believed to underlie these cardiovascular toxicities. nih.govjacc.org Clinical trials have reported instances of decreased LVEF, typically occurring within the first few months of treatment. europa.eunih.govjacc.org The incidence of reduced LVEF appears to be higher when this compound is used in combination with a BRAF inhibitor compared to BRAF inhibitor monotherapy. jacc.org While other kinase inhibitors are known to prolong the QT interval, a small study specifically evaluating this compound did not find significant QT prolongation. nih.gov The detailed mechanisms leading to this compound-induced cardiac dysfunction are still being elucidated through ongoing research. nih.gov
Hemorrhagic Event Mechanisms
Hemorrhagic events, ranging in severity and including fatal cases, have been reported in patients treated with this compound, particularly in the context of combination therapy with dabrafenib. cancercareontario.cabccancer.bc.caeuropa.eumedscape.comnih.govfda.gov The exact mechanisms responsible for these bleeding complications are not yet fully understood. nih.gov Concomitant use of antiplatelet or anticoagulant medications may increase the risk of hemorrhage. bccancer.bc.caeuropa.eu Fatal cerebral hemorrhages have been noted, especially in patients who develop brain metastases during combination treatment. cancercareontario.cabccancer.bc.ca
Dermatological Toxicities and Paradoxical MAPK Pathway Activation in Skin
Dermatological toxicities are frequently observed adverse effects of this compound. nih.govdovepress.comamegroups.orgactasdermo.org These reactions encompass various manifestations such as rash and acneiform dermatitis. nih.govdovepress.comamegroups.orgactasdermo.org While MEK inhibitors can cause skin toxicities that bear resemblance to those induced by EGFR inhibitors, the underlying mechanism differs from the paradoxical activation of the MAPK pathway seen with BRAF inhibitors in cells with wild-type BRAF. amegroups.orgactasdermo.orgeviq.org.aufrontiersin.orgdermnetnz.orgdovepress.com This paradoxical activation by BRAF inhibitors can lead to proliferative skin lesions like squamous cell carcinoma and keratoacanthoma. amegroups.orgeviq.org.aufrontiersin.orgdermnetnz.org In contrast, MEK inhibitors alone have generally not been associated with an increased incidence of these specific proliferative lesions. dovepress.comamegroups.org However, when this compound is combined with a BRAF inhibitor, the MEK inhibition helps to mitigate the occurrence of these proliferative skin toxicities by counteracting the paradoxical MAPK activation in wild-type BRAF keratinocytes. actasdermo.orgeviq.org.aufrontiersin.orgdermnetnz.org The mechanism behind acneiform eruptions with this compound might involve IGF-1-mediated sebaceous gland lipogenesis via the PI3K-AKT pathway or be related to an abrupt blockage of the MAPK pathway, similar to the effects seen with EGFR inhibitors. actasdermo.org
Ocular Manifestations (e.g., Retinal Vein Occlusion, Central Serous Retinopathy, Uveitis)
Ocular toxicities, including retinal vein occlusion (RVO), central serous retinopathy (CSR), retinal pigment epithelial detachment (RPED), and uveitis, have been reported in patients receiving this compound. bccancer.bc.caeuropa.eudovepress.comcancercareontario.camedscape.comnih.govkarger.comretinavitreus.commdpi.com The precise mechanisms underlying these ocular adverse events are not fully elucidated. nih.govkarger.com It has been proposed that inhibition of the MAPK pathway can induce an inflammatory response and compromise the blood-retinal barrier. karger.comretinavitreus.com Studies have also shown that MEK inhibition can directly lead to acute toxicity in retinal pigment epithelial cells, increasing permeability and disrupting the blood-retina barrier, which may contribute to the development of serous retinal detachment and macular edema. mdpi.com Uveitis and iridocyclitis have been observed, particularly in patients receiving combination therapy. europa.eukarger.comretinavitreus.com RVO is considered a serious complication that can potentially result in macular edema and vision loss. amegroups.org
Gastrointestinal Complications (e.g., Colitis, Perforation)
Gastrointestinal complications, such as colitis and gastrointestinal perforation, have been reported with this compound monotherapy and in combination with dabrafenib. bccancer.bc.caeuropa.eumedscape.comfda.govwww.gov.ukresearchsquare.com The inhibitory effects of this compound on angiogenesis and the proliferation of gastrointestinal epithelial cells may contribute to the risk of gastrointestinal perforation. www.gov.uk In patients with metastases in the gastrointestinal tract, rapid reduction in tumor size following combination therapy could also potentially lead to perforation at the metastatic sites. www.gov.uk Factors that may increase the risk of gastrointestinal perforation include a history of diverticulitis, presence of gastrointestinal metastases, and concurrent use of medications known to increase the risk of perforation. bccancer.bc.caeuropa.euwww.gov.uk
Musculoskeletal Effects (e.g., Rhabdomyolysis, Creatine Kinase Elevation)
Musculoskeletal adverse events, including myalgia, muscle weakness, and elevations in creatine kinase (CK), have been reported with this compound treatment. Rhabdomyolysis, a severe form of muscle breakdown characterized by significant CK elevation, has also been observed in patients receiving this compound as monotherapy or in combination with dabrafenib. europa.eucancercareontario.ca
Creatine kinase is an enzyme vital for cellular energy metabolism and muscle function, catalyzing the reversible transfer of phosphate groups between creatine and ATP to provide energy for muscle contraction and transport systems. nih.gov Elevated serum CK levels can result from various factors, including drug exposure. nih.gov While the precise mechanism by which this compound leads to CK elevation and rhabdomyolysis is not fully elucidated, it is understood that tyrosine kinases (TKs), which are inhibited by TK inhibitors like this compound, play roles in normal cellular functions, including muscle metabolism. nih.gov It is hypothesized that the inhibition of kinases within muscle tissue by this compound could inadvertently impact normal muscle activity, potentially leading to cellular stress or damage and subsequent CK release and muscle breakdown. nih.gov
Some cases of elevated CK levels have been noted without overt signs of rhabdomyolysis or other underlying muscle pathology. targetedonc.comnews-medical.net However, severe symptomatic CK elevation may necessitate dose reduction or treatment interruption, which could indirectly affect the efficacy of this compound. nih.gov A rare adverse effect reported with MEK inhibitors is dropped-head syndrome, characterized by progressive weakness of neck extensors due to focal non-inflammatory myopathy, often accompanied by high CK levels. news-medical.net This condition typically resolves upon discontinuation of the MEK inhibitor. news-medical.net
Potential Immune-Related Adverse Events
While immune checkpoint inhibitors are well-established for their association with immune-related adverse events (irAEs) due to their mechanism of activating the immune system, there is growing evidence suggesting that targeted therapies like BRAF and MEK inhibitors, including this compound, may also influence the immune system and potentially contribute to irAEs. spandidos-publications.comnih.govdermnetnz.orgoup.com
Preclinical and clinical studies indicate that BRAF and MEK inhibitors can modulate immune cell activation and the tumor microenvironment. spandidos-publications.comnih.govmdpi.com For instance, dabrafenib and this compound have been shown to enhance the expression of melanoma antigens on BRAF-mutated cells, potentially favoring an immune response against cancer cells. spandidos-publications.comnih.gov MEK inhibitors may also suppress PD-L1 expression on melanoma cells, making them more susceptible to immune destruction. spandidos-publications.com In murine models, this compound has been shown to decrease certain immunosuppression factors and increase CD4+ tumor-infiltrating lymphocyte levels. spandidos-publications.com When combined with anti-PD1 antibodies, it can also increase CD8+ cells. spandidos-publications.com
Despite these observations suggesting a potential for immune modulation, the effect of this compound on immune cells appears complex and context-dependent. In vitro studies have shown that this compound can reduce pERK levels in T cells and result in partial or transient inhibition of T-cell proliferation and alteration of cytokine production. mdpi.comaacrjournals.org It can also reduce the viability and proliferation of T-lymphocytes and production of IFNγ and other cytokines in vitro. nih.gov However, in vivo data from immunocompetent mouse models suggest that this compound may have minimal inhibitory effects on circulating immune cells and can even enhance the efficacy and/or tumor infiltration of CD8+ effector cells. aacrjournals.org
Cases of potential irAEs, such as sarcoidosis, mesenteric panniculitis, lymphocytic colitis, and neuropathy, have been reported in patients treated with the combination of dabrafenib and this compound. spandidos-publications.comnih.gov These occurrences suggest a possible role for BRAF and MEK inhibitors in triggering immune system activation and subsequent toxicities. spandidos-publications.comnih.gov It has been hypothesized that, similar to irAEs seen with immune checkpoint inhibitors which are mediated by the activity of CD4+ and CD8+ lymphocytes, the influence of dabrafenib and this compound on these cell types could be involved in the onset of such toxicities. spandidos-publications.comnih.gov The long-term efficacy of adjuvant MAPK inhibitors might even be partly attributable to their immunological function. frontiersin.org
Impact of Adverse Events on Treatment Adherence and Outcomes
Adverse events associated with this compound, particularly when used in combination with dabrafenib, can significantly impact patient medication adherence and, consequently, treatment outcomes. nih.govnih.gov The occurrence of toxicities, which can sometimes be complex and substantial, may lead to dose interruptions, dose reductions, or even permanent discontinuation of therapy. nih.govnih.govjhoponline.com
Poor adherence to oral anticancer drugs like this compound, often a consequence of side effects that negatively affect a patient's quality of life, can result in suboptimal drug exposure. nih.gov Significant interindividual variability in pharmacokinetics, influenced by factors such as age and body weight, further contributes to the risk of suboptimal exposure, potentially leading to treatment failure or increased toxicity. nih.gov
Studies have highlighted that managing adverse events effectively is crucial for maintaining treatment adherence and optimizing clinical outcomes. For example, in the COMBI-AD trial, pyrexia was a common AE associated with dabrafenib plus this compound, leading to drug discontinuation in a notable percentage of participants. jhoponline.com However, the implementation of improved pyrexia management algorithms, as demonstrated in the COMBI-APlus trial, has shown a reduction in the incidence of severe pyrexia and a lower treatment discontinuation rate, with comparable efficacy outcomes. jhoponline.com This underscores the importance of proactive AE management and patient support to ensure patients can remain on therapy and potentially achieve better long-term outcomes. nih.govnih.govjhoponline.com
Future Directions and Unanswered Research Questions
Elucidation of Novel Resistance Mechanisms
Acquired resistance to MEK inhibitors, including trametinib, is a significant challenge that limits the durability of response in patients. amsj.orgnih.gov While some resistance mechanisms involving alterations in BRAF, MEK1/2, and NRAS are known, novel and complex mechanisms continue to emerge. amsj.orgfrontiersin.org Future research aims to fully elucidate these diverse mechanisms to inform the development of strategies to overcome them. Studies are investigating alterations that reactivate the MAPK pathway downstream of MEK or activate alternative survival pathways. amsj.orgnih.gov For instance, increased expression of receptor tyrosine kinases (RTKs) like IGF-1R and PDGFR has been observed in some patients who develop resistance, suggesting the activation of the PI3K/AKT signaling pathway as a bypass mechanism. nih.gov Understanding these complex interactions and identifying the prevalence of various compensatory mechanisms in larger patient cohorts is crucial for designing effective next-generation therapies. nih.gov Research using preclinical models, such as cell lines derived from resistant tumors, and clinical data from patient samples are integral to this effort. nih.gov
Development of Predictive Biomarker Panels for Response and Resistance
Identifying biomarkers that can predict response to this compound or foretell the development of resistance is a key area of investigation. Currently, BRAF V600 mutations serve as a primary predictive marker for response to BRAF and MEK inhibitors. However, the heterogeneity of treatment outcomes highlights the need for more comprehensive predictive tools. mdpi.com Research is ongoing to identify dynamic biomarkers that reflect real-time tumor cell biology and can predict cellular outcomes like apoptosis, necrosis, senescence, or autophagy in response to this compound. nih.gov For example, studies in non-small cell lung cancer (NSCLC) cell lines suggest that cleaved PARP, phospho-4E-BP1, and phospho-AKT expression may predict these outcomes. nih.gov The development of panels incorporating multiple biomarkers, potentially including those related to the tumor microenvironment and immune system activation, could help tailor treatment strategies and identify patients most likely to benefit from this compound-based therapies or those who may require alternative or combination approaches from the outset. mdpi.comresearchgate.net
Optimization of Combination Therapy Regimens for Enhanced Efficacy and Reduced Toxicity
This compound is frequently used in combination with dabrafenib, a BRAF inhibitor, which has shown improved efficacy and reduced certain toxicities compared to single-agent BRAF inhibition. ascopubs.orgmednet.co.il However, the combination is associated with its own set of adverse events, such as pyrexia. mednet.co.iljhoponline.com Future research focuses on optimizing these combination regimens, including exploring novel combinations and strategies to manage toxicity while maintaining or enhancing efficacy. This includes investigating combinations with other targeted therapies, such as PI3K/AKT inhibitors, to overcome resistance mechanisms that involve the activation of parallel survival pathways. nih.govnih.gov Furthermore, combining this compound with immunotherapies, such as PD-1 or CTLA-4 inhibitors, is an active area of research, aiming to leverage complementary mechanisms to improve anti-tumor activity and overcome resistance. nih.govnih.govnih.govnih.gov Optimizing the sequence and timing of these combination therapies is also being explored to potentially reduce toxicity and enhance long-term outcomes. mdpi.com
Exploring this compound in Rare Mutations and Tumor Types
While this compound is established in BRAF V600-mutated melanoma, its potential utility in other cancers harboring BRAF mutations or other MAPK pathway alterations is being actively investigated. The Rare Oncology Agnostic Research (ROAR) trial and the NCI-MATCH study have explored the efficacy of dabrafenib and this compound in various rare cancers with BRAF V600E mutations, including anaplastic thyroid cancer, biliary tract cancer, and certain gliomas. ascopubs.orgmdpi.comascopubs.org These studies have shown promising results, leading to approvals in some of these rare tumor types. ascopubs.orgmdpi.comascopubs.orgmdanderson.org Future directions include further investigating this compound, alone or in combination, in other rare mutations within the MAPK pathway and in tumor types where this pathway is implicated, even in the absence of BRAF V600 mutations. jadpro.com This includes exploring its role in low-grade serous ovarian carcinoma, where activating mutations in the MAPK pathway are prevalent. mdanderson.orgonclive.com
Role of this compound in Adjuvant and Neoadjuvant Settings Beyond Approved Indications
The combination of dabrafenib and this compound has demonstrated significant benefit as adjuvant therapy in resected Stage III BRAF-mutated melanoma, improving relapse-free survival. targetedonc.comoncnursingnews.comcancernetwork.com Research is ongoing to explore the role of this compound, particularly in combination, in the adjuvant and neoadjuvant settings for other cancer types where the MAPK pathway is dysregulated. Studies are investigating neoadjuvant dabrafenib and this compound in high-risk resectable melanoma, showing promising pathological complete response rates and improved event-free survival compared to standard of care in some trials. ascopubs.orgfrontiersin.orgnih.gov Further research is needed to determine the optimal duration and patient selection for adjuvant and neoadjuvant this compound-based therapies in melanoma and to explore its potential utility in these settings for other malignancies. ascopubs.orgfrontiersin.orgnih.gov
Understanding Long-Term Clinical Outcomes and Mechanisms of Sustained Response
9.8. Pharmacogenomic and Real-World Evidence Studies to Inform Clinical Practice
Pharmacogenomic and real-world evidence (RWE) studies are increasingly important for understanding how genetic variations and clinical practice patterns influence patient outcomes with targeted therapies like this compound. These studies complement traditional randomized controlled trials (RCTs) by providing insights into the effectiveness and safety of treatments in broader, more diverse patient populations encountered in routine clinical care ispor.orgresearchgate.net.
Real-world data (RWD), collected from sources such as electronic health records, disease registries, and observational studies, are the foundation for generating RWE ispor.org. RWE can help address questions about treatment outcomes, disease progression, and comparisons between treatments where head-to-head RCTs may not be feasible ispor.org. For this compound, particularly in combination with dabrafenib, RWE studies have been conducted to evaluate its effectiveness in patient populations that may have been underrepresented in pivotal trials, such as those with brain metastases or previous treatments jhoponline.commdpi.com.
Another RWE study in Bulgaria assessed the effectiveness of dabrafenib and this compound in BRAF-positive melanoma patients treated in routine clinical practice from 2018 to 2022 pensoft.netresearchgate.net. This study compared real-world outcomes, including OS and PFS, against those reported in the COMBI-d and COMBI-v clinical trials pensoft.netresearchgate.net. The results suggested that real-world OS and PFS outcomes outperformed those from the COMBI-d trial pensoft.netresearchgate.net.
In non-small cell lung cancer (NSCLC) with BRAF V600E mutation, real-world data from the IFCT-2004 BLaDE study also supported the efficacy of dabrafenib-trametinib in both pretreated and untreated patients oncodaily.com. The study reported a 12-month OS rate of 67.4% and a median PFS of 10.4 months in second-line or later treatment and 18.2 months in first-line treatment oncodaily.com.
Pharmacogenomics, the study of how genetic makeup influences drug response, is another area contributing to personalized treatment approaches frontiersin.orgnih.govbps.ac.uk. In oncology, pharmacogenomic studies investigate both somatic mutations within tumors and germline variants in the patient's DNA that may affect drug efficacy or toxicity frontiersin.orgpharmgkb.orgijmio.com. For targeted therapies like this compound, which acts on the MAPK pathway, understanding the pharmacogenomic landscape can help predict response and identify potential resistance mechanisms mdpi.commdpi.com.
Research using cancer cell lines has aimed to identify this compound response markers mdpi.com. For example, studies have explored the association between specific gene alterations, such as CDKN2A copy number loss in sensitive cell lines and FLOT1 overexpression in resistant cell lines, and response to this compound in glioblastoma multiforme cell lines mdpi.com. While pharmacogenomic studies have identified potential markers, translating these findings into routine clinical practice faces challenges, including the need for robust clinical validation and integration into clinical decision support systems frontiersin.orgnih.gov.
Real-world evidence and pharmacogenomic studies are crucial for refining the use of this compound and informing clinical practice by providing data on diverse patient populations and exploring the genetic basis of response and resistance.
Here is a summary of some real-world effectiveness data for dabrafenib plus this compound:
| Study (Real-World) | Patient Population | N | Median PFS (months) | Median OS (months) | Objective Response Rate (%) |
| COMBI-r (Melanoma) | BRAF V600-mutated, Stage IV (Overall) | 273 | - | - | - |
| COMBI-r (Melanoma) | Stage IV with Brain Metastases | 100 | 6.1 | 11.6 | 31.9 |
| COMBI-r (Melanoma) | Stage IV without Brain Metastases | 173 | 10.5 | 21.7 | 44.5 |
| Bulgarian RWE | BRAF-positive Melanoma | 190 | Outperformed COMBI-d | Outperformed COMBI-d | - |
| IFCT-2004 BLaDE | BRAF V600E-mutated NSCLC (2L+) | - | 10.4 | 67.4% (12-month OS rate) | Significant efficacy |
| IFCT-2004 BLaDE | BRAF V600E-mutated NSCLC (1L) | - | 18.2 | - | Significant efficacy |
*Outcomes in the Bulgarian RWE study were reported as outperforming those in the COMBI-d trial, which had a median PFS of 9.3 months and median OS of 25.1 months pensoft.net.
Q & A
Q. What are the primary molecular mechanisms of Trametinib, and how can researchers experimentally validate its target specificity?
this compound is a selective MEK1/MEK2 inhibitor that blocks the MAPK pathway, critical in cancer cell proliferation. To validate target specificity:
- Methodology : Use X-ray crystallography to resolve this compound-MEK-KSR complexes, confirming binding interfaces (e.g., residues involved in hydrogen bonding or hydrophobic interactions) .
- Functional assays : Measure MAPK pathway inhibition via Western blotting for phosphorylated ERK (p-ERK) in cell lines (e.g., BRAF V600E-mutant melanoma models) .
- Controls : Include MEK-negative cell lines or CRISPR-Cas9 MEK-knockout models to rule off-target effects.
Q. How should researchers design in vitro experiments to assess this compound’s pharmacokinetic properties?
- Key parameters : Solubility (via HPLC), plasma protein binding (equilibrium dialysis), and metabolic stability (using liver microsomes) .
- Methodology :
- Accelerator Mass Spectrometry (AMS) : Quantify [14C]-Trametinib in plasma to track absorption and distribution .
- Caco-2 assays : Evaluate intestinal permeability to predict oral bioavailability.
- Data interpretation : Compare results to known MEK inhibitors (e.g., Cobimetinib) to contextualize this compound’s unique properties.
Q. What are the standard protocols for synthesizing and characterizing this compound in preclinical studies?
- Synthesis : Follow routes described in patent literature, starting from 2-fluoro-4-iodoaniline and other intermediates. Purify via column chromatography .
- Characterization :
- Spectroscopy : NMR (¹H, 13C) and high-resolution mass spectrometry (HRMS) for structural confirmation.
- Purity analysis : Use HPLC with UV detection (λ = 254 nm) and quantify impurities (e.g., process-related byproducts) .
Advanced Research Questions
Q. How can researchers optimize this compound combination therapies to overcome resistance in BRAF-mutant cancers?
- Experimental design :
- Synergy quantification : Apply the Chou-Talalay method to calculate Combination Index (CI) values. For example, combine this compound with BRAF inhibitors (e.g., Dabrafenib) at varying ratios in 3D spheroid models .
- Resistance models : Generate this compound-resistant cell lines via chronic exposure and profile mutations (e.g., MEK1 Q56P) using whole-exome sequencing.
- Data analysis : Use isobolograms to visualize synergistic regions (CI < 1) and validate in vivo using PDX models .
Q. How should contradictory findings in this compound clinical trial data be analyzed?
- Case example : Discrepancies in progression-free survival (PFS) across trials may arise from patient stratification (e.g., BRAF mutation subtypes).
- Methodology :
- Reporting : Follow PRISMA guidelines and disclose conflicts of interest in systematic reviews .
Q. What advanced techniques are used to study this compound’s off-target effects in non-cancerous tissues?
- Methodology :
- Proteome-wide profiling : Use affinity pulldown assays with this compound-conjugated beads to identify unintended kinase targets .
- Single-cell RNA sequencing : Compare transcriptional profiles of treated vs. untreated normal tissues (e.g., cardiac or hepatic cells) to detect toxicity signatures.
- Validation : Knock down candidate off-target kinases (e.g., FLT3) and reassess this compound’s cytotoxicity .
Methodological Guidance for Experimental Design
Q. How to design a Phase I/II clinical trial evaluating this compound in novel cancer indications?
- Phase I : Use a 3+3 dose-escalation design to determine MTD. Key endpoints: Dose-limiting toxicities (e.g., pyrexia, rash) and pharmacokinetics (Cmax, AUC) .
- Phase II : Stratify patients by biomarker status (e.g., BRAF V600E/K vs. non-V600). Use RECIST 1.1 criteria for response evaluation .
- Data collection : Standardize adverse event reporting using CTCAE v5.0 and archive biospecimens for correlative studies (e.g., phospho-ERK levels) .
Q. What statistical approaches are recommended for analyzing this compound’s long-term survival data?
- Survival analysis : Use Cox proportional hazards models to adjust for covariates (e.g., age, LDH levels). Report hazard ratios (HR) with 95% confidence intervals .
- Handling censored data : Apply Kaplan-Meier estimates for PFS and overall survival (OS), and compare curves via log-rank tests .
Data Presentation and Reproducibility
Q. How to ensure reproducibility in this compound preclinical studies?
- Documentation : Provide detailed synthesis protocols (e.g., solvent ratios, reaction temperatures) in Supplementary Materials .
- Data sharing : Deposit raw proteomics/sequencing data in public repositories (e.g., GEO, PRIDE) with unique accession numbers .
- Reagent validation : Authenticate cell lines via STR profiling and confirm MEK inhibitor activity in each batch using p-ERK assays .
Tables for Key Findings
Table 1 : Efficacy of this compound + Dabrafenib in BRAF V600E Melanoma
| Endpoint | Combination (150/2 mg) | Monotherapy (150 mg) | P-value |
|---|---|---|---|
| Median PFS (months) | 9.4 | 5.8 | <0.001 |
| Objective Response Rate | 76% | 54% | 0.03 |
| Cutaneous SCC Incidence | 7% | 19% | 0.09 |
Table 2 : Key Physicochemical Properties of this compound
| Property | Value |
|---|---|
| Molecular Formula | C26H23FIN5O4 |
| Molecular Weight | 615.4 g/mol |
| Solubility (pH 7.4) | 0.02 mg/mL (DMSO-soluble) |
| Plasma Protein Binding | 97.4% |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Featured Recommendations
| Most viewed | ||
|---|---|---|
| Most popular with customers |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.


