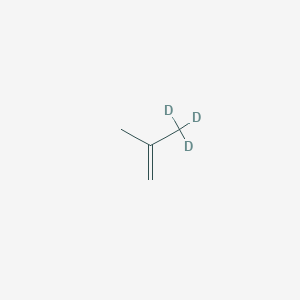
3,3,3-Trideuterio-2-methylprop-1-ene
Overview
Description
3,3,3-Trideuterio-2-methylprop-1-ene (C₄H₃D₃) is a deuterated derivative of 2-methylprop-1-ene (isobutylene), where three hydrogen atoms on the terminal methyl group are replaced by deuterium. This isotopic substitution significantly impacts its physical and chemical properties, particularly in kinetic isotope effects (KIEs) and spectroscopic behavior. While the compound is structurally analogous to non-deuterated isobutylene, its deuteration enhances stability in radical reactions and reduces reaction rates in processes involving C-H(D) bond cleavage. Applications include its use as a tracer in mechanistic studies and in synthetic chemistry to probe reaction pathways .
Preparation Methods
Synthetic Routes and Reaction Conditions
The synthesis of 3,3,3-Trideuterio-2-methylprop-1-ene typically involves the deuteration of 2-methylpropene. This process can be achieved through the catalytic exchange of hydrogen atoms with deuterium atoms using deuterium gas (D2) in the presence of a suitable catalyst, such as palladium on carbon (Pd/C). The reaction conditions often include elevated temperatures and pressures to facilitate the exchange process.
Industrial Production Methods
In an industrial setting, the production of this compound may involve continuous flow reactors to ensure efficient deuteration. The use of deuterium gas in large quantities and the recycling of unreacted deuterium are common practices to optimize the yield and reduce costs.
Chemical Reactions Analysis
Types of Reactions
3,3,3-Trideuterio-2-methylprop-1-ene undergoes various chemical reactions, including:
Oxidation: The compound can be oxidized to form corresponding alcohols, aldehydes, or carboxylic acids.
Reduction: Reduction reactions can convert it into saturated hydrocarbons.
Substitution: It can participate in substitution reactions where deuterium atoms are replaced by other functional groups.
Common Reagents and Conditions
Oxidation: Common oxidizing agents include potassium permanganate (KMnO4) and chromium trioxide (CrO3).
Reduction: Catalytic hydrogenation using hydrogen gas (H2) and a catalyst such as palladium on carbon (Pd/C).
Substitution: Halogenation reactions using halogens like chlorine (Cl2) or bromine (Br2) under controlled conditions.
Major Products
Oxidation: Formation of deuterated alcohols, aldehydes, or carboxylic acids.
Reduction: Formation of deuterated alkanes.
Substitution: Formation of deuterated halides.
Scientific Research Applications
Chemistry
In the field of chemistry, 3,3,3-Trideuterio-2-methylprop-1-ene serves as a vital tool in nuclear magnetic resonance (NMR) spectroscopy. Its deuterated nature allows for clearer spectra due to reduced background noise from hydrogen signals. This property is particularly useful in studying complex reaction mechanisms and molecular structures.
Biology
The compound is utilized as a tracer in metabolic studies. By replacing hydrogen with deuterium, researchers can track the metabolic pathways of substances in biological systems. This application is critical for understanding biochemical processes and drug metabolism.
Medicine
In pharmaceutical research, deuterated compounds like this compound are being explored for their potential to enhance the pharmacokinetic properties of drugs. Deuteration can improve the stability and reduce the metabolic rate of certain drugs, leading to prolonged efficacy in therapeutic applications.
Industry
Industrially, this compound is employed in the production of deuterated polymers and materials. These materials exhibit unique physical and chemical properties that can be advantageous in various applications including advanced coatings and specialty chemicals.
Case Studies
Case Study 1: NMR Spectroscopy
In a study published in Journal of Organic Chemistry, researchers used this compound to investigate the kinetics of a reaction involving free radicals. The deuterated compound provided clearer NMR signals that allowed for more accurate kinetic measurements.
Case Study 2: Metabolic Tracing
A metabolic tracing study conducted at a university utilized this compound to track the pathways of lipid metabolism in mammals. The results indicated that deuterated compounds could significantly alter metabolic rates compared to their non-deuterated counterparts.
Case Study 3: Drug Development
Research on a new class of anti-inflammatory drugs incorporated this compound to enhance drug stability. The findings showed improved half-life and bioavailability compared to traditional formulations without deuteration.
Mechanism of Action
The mechanism by which 3,3,3-Trideuterio-2-methylprop-1-ene exerts its effects involves the replacement of hydrogen atoms with deuterium atoms. This isotopic substitution can alter the compound’s physical and chemical properties, such as bond strength and reaction rates. The molecular targets and pathways involved depend on the specific application, such as its role as a tracer in metabolic studies or its use in NMR spectroscopy.
Comparison with Similar Compounds
Comparison with Structurally Similar Compounds
Ethyl 3,3-Difluoro-2-(trifluoromethyl)prop-2-enoate
Structure : C₆H₅F₅O₂ (CAS 41296-90-6).
Key Differences :
- Substituents : Contains fluorine atoms (electron-withdrawing groups) instead of deuterium.
- Reactivity : Fluorine substituents increase polarity and electrophilicity, making it reactive in cycloaddition and nucleophilic substitution reactions.
- Applications : Used in fluoropolymer synthesis and agrochemical intermediates.
3,3-Diethoxy-1-Iodo-2-methylprop-1-ene
Structure : C₈H₁₅IO₂ (CAS 61147-90-8).
Key Differences :
- Substituents : Features ethoxy and iodine groups, introducing steric bulk and electrophilic character.
- Reactivity : Iodine enhances susceptibility to nucleophilic attack, while ethoxy groups stabilize the alkene via resonance.
- Applications: Intermediate in cross-coupling reactions and organoiodine chemistry.
1,2,3-Trichloropropane
Structure : C₃H₅Cl₃ (CAS 96-18-4).
Key Differences :
- Substituents : Chlorine atoms replace deuterium and methyl groups.
- Reactivity : Chlorine imparts toxicity and environmental persistence. Reacts via SN mechanisms.
- Applications: Historical use as a solvent (now restricted due to toxicity).
| Property | This compound | 1,2,3-Trichloropropane |
|---|---|---|
| Molecular Weight (g/mol) | ~70.11 (estimated) | 147.43 |
| Toxicity | Low | High (carcinogenic) |
| Environmental Impact | Negligible | Persistent pollutant |
Research Findings and Implications
- Kinetic Isotope Effects: The deuterated compound exhibits a 5–10× slower reaction rate in hydrogen-abstraction reactions compared to non-deuterated analogs, critical for studying enzyme mechanisms .
- Synthetic Utility: Ethyl 3,3-difluoro-2-(trifluoromethyl)prop-2-enoate’s fluorinated structure enables unique regioselectivity in Diels-Alder reactions .
- Safety Profile : Unlike 1,2,3-trichloropropane, this compound poses minimal environmental or health risks, making it preferable for lab use .
Biological Activity
Overview
3,3,3-Trideuterio-2-methylprop-1-ene is a deuterated alkene that serves various industrial applications, including the production of synthetic rubber, plastics, and fuel additives. Its unique isotopic composition makes it particularly useful in scientific research, especially in areas such as nuclear magnetic resonance (NMR) spectroscopy and metabolic studies. This article explores the biological activity of this compound, highlighting its mechanisms of action, potential therapeutic applications, and relevant research findings.
| Property | Value |
|---|---|
| CAS Number | 110597-10-9 |
| Molecular Formula | CHD |
| Molecular Weight | 89.14 g/mol |
| IUPAC Name | This compound |
Synthesis and Preparation
The synthesis of this compound typically involves the deuteration of 2-methylpropene through catalytic exchange with deuterium gas (D). This process often utilizes catalysts like palladium on carbon (Pd/C) under elevated temperatures and pressures to facilitate the reaction. In industrial settings, continuous flow reactors are employed to optimize yield and reduce costs associated with deuterium usage.
The biological activity of this compound is primarily attributed to its isotopic substitution of hydrogen atoms with deuterium. This alteration can influence the compound's physical and chemical properties, including bond strength and reaction rates. The specific pathways and molecular targets involved depend on its application:
- NMR Spectroscopy : The presence of deuterium enhances spectral resolution and sensitivity in NMR studies.
- Metabolic Tracing : It serves as a tracer in metabolic studies to elucidate biochemical pathways.
Research Findings
Recent studies have highlighted the potential biological activities of this compound:
- Antioxidant Properties : Research indicates that deuterated compounds can exhibit enhanced antioxidant activity compared to their non-deuterated counterparts. This property is crucial for applications in preventing oxidative stress-related diseases.
- Pharmacokinetics : Deuterated drugs are being explored for their improved pharmacokinetic properties. The incorporation of deuterium can lead to slower metabolic degradation, potentially enhancing the efficacy and duration of action of therapeutic agents.
- Metabolic Studies : In metabolic tracing experiments, this compound has been used to study various biochemical pathways. Its unique isotopic signature allows for precise tracking within complex biological systems.
Case Study 1: NMR Applications
In a study focusing on reaction mechanisms within organic compounds, this compound was utilized as a model compound in NMR spectroscopy. The results demonstrated that the presence of deuterium significantly improved the clarity of spectral data, allowing researchers to deduce reaction pathways more accurately.
Case Study 2: Metabolic Pathway Tracing
Another study investigated the use of this compound as a tracer in metabolic pathways involving lipid metabolism. By substituting hydrogen with deuterium in fatty acids, researchers were able to trace the incorporation of these lipids into cellular structures more effectively than with non-deuterated analogs.
Q & A
Basic Research Questions
Q. What synthetic routes are most effective for producing 3,3,3-Trideuterio-2-methylprop-1-ene with high isotopic purity?
- Methodological Answer : Deuteration can be achieved via catalytic exchange reactions using deuterated solvents (e.g., D₂O or CD₃OD) or hydrogen-deuterium (H-D) exchange catalysts like palladium or platinum oxides. Isotopic purity is optimized by iterative purification steps, such as fractional distillation or column chromatography, followed by validation via mass spectrometry (MS) and nuclear magnetic resonance (NMR) spectroscopy. For analogs, database searches (e.g., Cambridge Structural Database) highlight structural precedents for deuteration strategies .
Q. How can researchers confirm the structural integrity of this compound post-synthesis?
- Methodological Answer : Multinuclear NMR (¹H, ¹³C, ²H) is critical for verifying deuterium incorporation and positional fidelity. Gas chromatography-mass spectrometry (GC-MS) quantifies isotopic abundance, while infrared (IR) spectroscopy identifies functional group retention. Cross-referencing with structural analogs in crystallographic databases ensures conformational accuracy .
Advanced Research Questions
Q. What kinetic isotope effects (KIEs) are observed in reactions involving this compound, and how do they inform mechanistic studies?
- Methodological Answer : KIEs are measured via competitive experiments under controlled conditions (e.g., radical or electrophilic addition reactions). Deuterium substitution at the 3-position alters reaction rates, which are quantified using Arrhenius plots or computational modeling (DFT or MD simulations). Discrepancies between experimental and theoretical KIEs may indicate tunneling effects or transition-state modifications .
Q. How does isotopic labeling of this compound enhance metabolic or environmental tracer studies?
- Methodological Answer : Deuterated analogs serve as stable isotopic tracers in mass spectrometry imaging (MSI) or isotope-ratio mass spectrometry (IRMS) to track metabolic pathways or environmental degradation. Experimental designs must account for deuterium’s lower kinetic energy in bond cleavage compared to protium, which affects fragmentation patterns in MS .
Q. What computational models best predict the thermodynamic stability and reactivity of deuterated alkenes like this compound?
- Methodological Answer : Density functional theory (DFT) calculations with deuterium-specific basis sets (e.g., cc-pVTZ-D) model bond dissociation energies and reaction pathways. Molecular dynamics (MD) simulations assess solvent effects on deuteration efficiency. Validation requires correlation with experimental data, such as NMR chemical shifts or kinetic parameters .
Methodological and Data Analysis Questions
Q. How should researchers address contradictions in isotopic labeling efficiency between theoretical and experimental results?
- Methodological Answer : Systematic error analysis is essential. Replicate experiments under varying conditions (temperature, solvent, catalyst loading) to identify outliers. Use control compounds to isolate variables. Computational refinements, such as adjusting solvation models or vibrational frequencies in DFT, can reconcile discrepancies .
Q. What protocols ensure safe handling and disposal of deuterated compounds like this compound?
- Methodological Answer : Follow institutional guidelines for deuterated waste segregation. Use fume hoods and personal protective equipment (PPE) to minimize inhalation/contact. Collaborate with certified waste management services for deuterium-labeled compound disposal, as improper handling risks environmental isotopic contamination .
Q. Experimental Design and Reproducibility
Q. What steps improve reproducibility in deuteration reactions for this compound synthesis?
- Methodological Answer : Standardize catalyst activation (e.g., pre-reduction of Pd/C in D₂ atmosphere) and solvent deuteration levels. Document reaction parameters (e.g., stirring rate, degassing cycles) meticulously. Use internal standards (e.g., deuterated benzene) for NMR calibration to ensure consistent purity assessments .
Q. How can researchers design kinetic studies to isolate the effects of deuterium substitution in this compound?
- Methodological Answer : Employ pseudo-first-order conditions with excess reagents to minimize side reactions. Compare rate constants (k_H vs. k_D) using stopped-flow techniques or in-situ monitoring (e.g., FTIR). Statistical analysis (ANOVA) identifies significant deviations attributable to isotopic substitution .
Q. Structural and Functional Analysis
Q. What challenges arise in crystallographic characterization of deuterated alkenes, and how are they mitigated?
- Methodological Answer : Deuteration reduces X-ray scattering contrast, complicating electron density mapping. Neutron diffraction or high-resolution synchrotron X-ray sources enhance detection. Co-crystallization with heavy-atom derivatives (e.g., brominated hosts) improves phase resolution .
Properties
IUPAC Name |
3,3,3-trideuterio-2-methylprop-1-ene | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C4H8/c1-4(2)3/h1H2,2-3H3/i2D3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
VQTUBCCKSQIDNK-BMSJAHLVSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(=C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
[2H]C([2H])([2H])C(=C)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C4H8 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
59.12 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details





Synthesis routes and methods III
Procedure details











Synthesis routes and methods IV
Procedure details









Synthesis routes and methods V
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.